The protein TAZ sends 'mixed signals' to stem cells
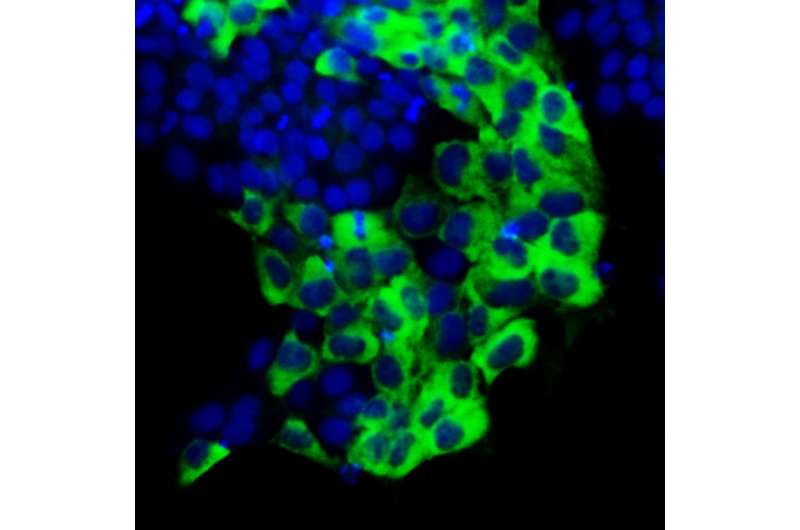
Just as beauty exists in the eye of the beholder, a signal depends upon the interpretation of the receiver. According to new USC research published in Stem Cell Reports, a protein called TAZ can convey very different signals—depending upon not only which variety of stem cell, but also which part of the stem cell receives it.
When it comes to varieties, some stem cells are "naïve" blank slates; others are "primed" to differentiate into certain types of more specialized cells. Among the truly naïve are mouse embryonic stem cells (ESCs), while the primed variety includes the slightly more differentiated mouse epiblast stem cells (EpiSCs) as well as so-called human "ESCs"—which may not be true ESCs at all.
In the new study, PhD student Xingliang Zhou and colleagues in the laboratory of Qi-Long Ying demonstrated that naïve mouse ESCs don't require TAZ in order to self-renew and produce more stem cells. However, they do need TAZ in order to differentiate into mouse EpiSCs.
The scientists observed an even more nuanced situation for the primed varieties of stem cells: mouse EpiSCs and human ESCs. When TAZ is located in the nucleus, this prompts primed stem cells to differentiate into more specialized cell types—a response similar to that of the naïve cells. However, if TAZ is in the cytoplasm, or the region between the nucleus and outer membrane, primed stem cells have the opposite reaction: they self-renew.
"TAZ has stirred up a lot of controversy in our field, because it appears to produce diverse and sometimes opposite effects in pluripotent stem cells," said Ying, senior author and associate professor of stem cell biology and regenerative medicine. "It turns out that TAZ can indeed produce opposite effects, depending upon both its subcellular location and the cell type in question."
First author Zhou added: "TAZ provides a new tool to stimulate stem cells to either differentiate or self-renew. This could have important regenerative medicine applications, including the development of a better way to generate the desired cell types for cell replacement therapy."
More information: Xingliang Zhou et al, Cytoplasmic and Nuclear TAZ Exert Distinct Functions in Regulating Primed Pluripotency, Stem Cell Reports (2017). DOI: 10.1016/j.stemcr.2017.07.019
Journal information: Stem Cell Reports
Provided by University of Southern California