Coal-burning power stations are becoming more environmentally friendly

In the course of Project SCARLET, scientists at the TU Darmstadt have succeeded in developing the so-called Carbonate Looping process for the reduction of CO2 emissions during power plant operations almost to the point of market readiness.
By the time Professor Bernd Epple of the TU Darmstadt's Institute for Energy Systems and Energy Technology opens the final symposium of the European-wide Project SCARLET these coming days, several milestones will have been reached. Over the past three years, Professor Epple, a mechanical engineer, and his ten German and international project partners have developed the technical prerequisites for the industrial deployment of the Carbonate Looping process. This process can be used to separate more than 90 per cent of the CO2 released during the combustion of fossil fuels. Retrofitting existing power generation facilities and industrial plants with this technology would allow them to be operated in a much more environmentally-friendly manner.
Based on data measured at a 1 Megawatt (MW) experimental facility, Professor Epple and his team have developed scaling tools for industrial plants with which they have conducted model calculations and computer simulations. They have proven that the new process is more cost effective and energy efficient than competing processes. They presented plans for a complete pilot project plant for the coal-fired power plant Émile Huchet in Saint-Avold in France, with a total output of 20 MW.
"If the funding details were already clarified at this point", says Dr. Jochen Ströhle, Senior Research Scientist at the Institute for Energy Systems and Technology at the TU Darmstadt and Project SCARLET coordinator, "then this could be the first industrial scale pilot project to be launched. "All the necessary planning, including a cost schedule and a risk assessment are already on the table". The acronym "SCARLET" stands for "Scale up of Calcium Carbonate Looping Technology for Efficient CO2 Capture from Power and Industrial Plants". The European Union has provided five million euro in project funding, and the overall budget was over seven million euro.
How does the carbonate looping process work?
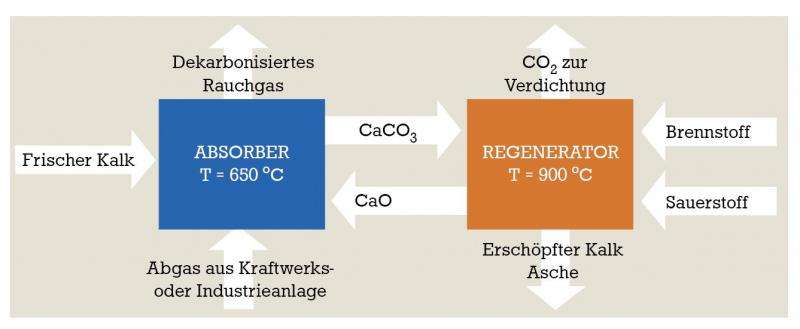
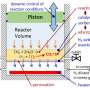
Carbonate Looping involves two chemical reactions continuously running in two interconnected fluidized bed reactors. In the first fluidized bed reactor, the absorber, a powder of burnt lime or calcium oxide (CaO) is contacted with the CO2 in the power station exhaust to form calcium carbonate (CaCO3). The calcium carbonate then is transferred in the second of the two fluidized bed reactors, known as the regenerator, where high temperatures force out the CO2 bound as the calcium carbonate thereby producing burnt lime and CO2 once again. The released CO2 can then be used for other purposes or stored whilst the burnt lime is returned to the first fluidized bed reactor. The material needs to be replaced after several dozen cycles, but the depleted lime can then be used in cement production making it a valuable raw material rather than a waste product.
"We need 80 kilograms of lime to capture one tonne of CO2", Professor Epple explains. "The current process costs amount to around 20 to 27 euro per tonne of CO2. Other processes are more expensive and less efficient. Through SCARLET we have succeeded in moving the Carbonate Looping process a major step forward towards market readiness".
What did the project partners do in particular? First, it was necessary to discover the conditions and prerequisites for the continuous operation of the 1 MW pilot plant within grounds of the TU Darmstadt. Only under stable operating conditions it is possible to extrapolate from the measured data to calculate the necessary scaling factors. The pilot plant was built six years ago with funding from the German Federal Ministry for Economic Affairs, the European Union, and various industrial partners. "Today, we operate the 1 MW facility for several weeks at a time with a constant level of CO2 sequestration", explains Jochen Hilz, a doctoral candidate who has been supporting the project right from the outset. "The reactivity of the lime declines as the number of cycles increases, which necessitates operating periods lasting up to several days, to be able to draw reliable conclusions to its long-term behaviour. We now know the conditions necessary for steady operations. The more empirical data we have from continuous operations, the fewer assumptions we need to make and the better are models and computer simulations that we calculate."
For SCARLET, it was necessary to upgrade the 1 MW experimental facility. "For example, we built a silo for 30 tonnes of fuel", Professor Epple reports, who developed the concepts for the process based on his professional work for an industrial corporation. "You see, we need a constant supply for continuous operation. We also had to improve the transfer of particles from one fluidized bed reactor to the other in addition to adapting existing measurement systems. What we wanted to know was how the particles move inside the fluidized bed reactors; their relative densities in various zones, and what velocities they can achieve. Also", Epple continues, "we wanted to analyse the gas at the different levels. That's the only way we can understand how fast the CO2 has been separated in the absorberand then set free again in the regenerator". Because temperatures of 900 degree Celsius are reached within the fluidized bed reactors, measuring this data is anything but trivial. "Basically", Epple explains, the "reactor contents are like molten lava".
It was not easy to measure what actually happens within the reactors during the Carbonate Looping process. As Dr. Ströhle explains: "We knew how the process behaves in general, and that high CO2 absorption efficiencies are possible. On the other hand though, we knew very little about the long-term behaviour of the lime under real operating conditions. That has now changed following SCARLET." This knowledge was used in collaboration with the project partners to develop models for larger facilities and various plant types. The process can also be used for industrial cement and steel plants as well as waste incineration facilities.
So, what's next? "We'll be taking various approaches" says Professor Epple, "Including, for example, planning a facility that separates out the CO2 produced during waste incineration." The project team is also discussing concepts for the utilization of the recovered CO2. The price per tonne of CO2 is currently very low on the global markets. At this point, any power plant operators making use of the process would not be in a position to be able to sell the captured CO2 at a rate that would enable them to break even, let alone make a profit. It would have to be stored or reprocessed. "We're considering using the CO2 directly for the production of methanol", says Professor Epple. "Global market prices for methanol are higher than those for CO2." He and his team have developed the process even further to the point where pure oxygen will no longer be required for combustion within the regenerator and the overall efficiency level will be increased. They are determined to bring the project to a successful conclusion.
Provided by Technische Universitat Darmstadt