A new approach to improving lithium-sulfur batteries

Rechargeable lithium-ion batteries are the power behind most modern portable electronics, including cell phones, tablets, laptops, fitness trackers, and smart watches. However, their energy density—that is, the amount of energy stored within a given amount of physical space, or mass—will need to be improved for these batteries to see widespread use in smart grid and electric transport applications.
In contrast, the energy density of lithium-sulfur (Li-S) batteries is five times higher than that of Li-ion batteries. That advantage, combined with low cost, suggests that this alternative technology shows promise for high-energy storage applications.
But the use of Li-S batteries is limited by a different problem: rapid capacity fade, which means that the amount of charge these batteries can deliver at the rated voltage decreases significantly with use.
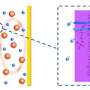
Bingqing Wei, professor in the Department of Mechanical Engineering at the University of Delaware, explains that this problem stems from a phenomenon known as the polysulfide shuttle effect, in which the spontaneous formation of polysulfides inhibits performance.
Now, Wei and colleagues have demonstrated a new polysulfide entrapping strategy that greatly improves the cycle stability of Li-S batteries.
The work is reported in the scientific article "Ferroelectric-Enhanced Polysulfide Trapping for Lithium-Sulfur Battery Improvement" published recently in Advanced Materials. The authors include researchers from Northwestern Polytechnical University, Shenzhen University and Hong Kong Polytechnic University in China.
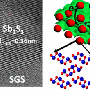
Wei explains that the addition of ferroelectric nanoparticles into the battery cathode anchors the polysulfides, preventing them from dissolving and causing the loss of active materials at the cathode.
"While the mechanism underlying the trapping of polysulfides is unclear at this point, we're optimistic about the potential of this approach to high-performance lithium-sulfur battery applications, as it not only solves the problem of the polysulfide shuttle effect but also can be seamlessly coupled to current industrial battery manufacturing processes," Wei says.
More information: Keyu Xie et al, Ferroelectric-Enhanced Polysulfide Trapping for Lithium-Sulfur Battery Improvement, Advanced Materials (2017). DOI: 10.1002/adma.201604724
Journal information: Advanced Materials
Provided by University of Delaware