Scientists build ion-selective membrane for ultra-stable lithium sulfur batteries
Advanced energy storage systems are highly desired to fill the gap between currently available battery systems and high performance electronic devices or even electric vehicles. As the commonly-used lithium ion battery systems are approaching their theoretical energy density value, lithium-sulfur batteries are considered to be one promising candidate, exhibiting much higher theoretical energy density at 2600 Wh/kg (around 3-5 times that of the lithium ion batteries). However, the practical applications of lithium-sulfur batteries are hindered by the complexity of this electrochemical system, especially the insulate nature of sulfur and the so called "shuttle effect", which means the diffusion and reaction of the cathode intermediate polysulfide with the anode side.
Researchers from Tsinghua University in Beijing, led by professors Qiang Zhang and Fei Wei, have developed a new strategy to build ultra-stable lithium-sulfur batteries based on an ion selective membrane system. With this new membrane system, the cyclic degradation of the cell was significantly reduced to 0.08 % per cycle within the first 500 cycles. Meanwhile, the coulombic efficiency of the battery can also be improved by around 10 %, which may greatly benefit the energy efficiency of the battery system. The team has published their findings in a recent issue of Energy & Environment Science.
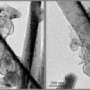
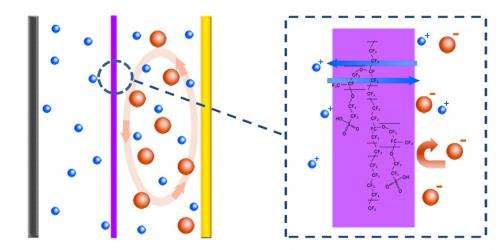
"Designs for cathode electrode structures for a lithium sulfur battery have been widely investigated, but a design to suppress the shuttle effect based on the whole battery system has rarely been reported," Qiang Zhang said. "We employ a cation permselective membrane, which helped to 'separate' lithium ions and polysulfide ions based on their different charge nature. The electrostatic interaction allows diffusion across the membrane but prevents the permeation of polysulfide anions, which suppressed the shuttle effect." In their experiments, a facile coating method was employed to build a complete selective ion shield between the cathode and anode electrode. By using a visualized glass cell, one can clearly observe that the polysulfide was prevented from reaching the anode side when using the ion selective membrane.
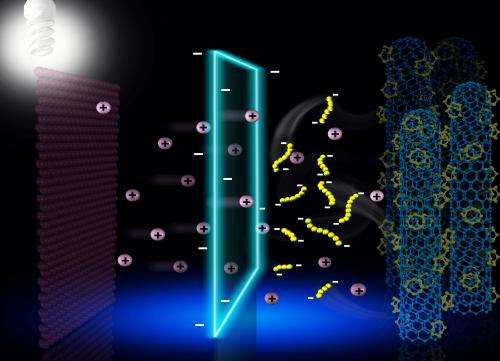
As Prof. Zhang and Wei point out, this approach shed some light on building ultra-stable lithium-sulfur batteries by suppressing the "shuttle effect". This method is also fully applicable with other advanced electrodes. Going forward, the researchers hope to understand the other problems in the degradation of lithium sulfur cells and to build even better batteries.
More information: Ionic shield for polysulfides towards highly-stable lithium-sulfur batteries, Energy & Environment Science.
Journal information: Energy & Environment Science
Provided by Tsinghua University