Balancing the 'yin' and 'yang' in lithium sulfur batteries
This decade has witnessed a rapid development in electric vehicles and portable electronics, accompanied by ever-increasing research enthusiasm for high-energy-density rechargeable batteries with long service life. Conventional Li-ion batteries are now approaching their theoretical energy density limits and cannot meet the practical demands of emerging markets. Scientists and engineers are therefore exploring better battery design and solutions.
"The lithium sulfur battery is recognized as a promising alternative for its intercalation chemistry counterparts," said Dr. Qiang Zhang, a faculty at Department of Chemical Engineering, Tsinghua University. "It possesses a theoretical energy density of ~2600 Wh kg-1 and provides a theoretical capacity of 1672 mAh g−1 through multi-electron redox reactions. Additionally, valuable characteristics like high natural abundance, low cost and environmental friendliness of sulfur have lent competitive edges to the lithium sulfur battery."
In commonly used liquid electrolytes for lithium sulfur batteries, the lithiation of sulfur involves multiphase solid-liquid-solid conversion. The highly soluble intermediates, known as lithium polysulfides, are prone to itinerate between sulfur cathode and lithium anode, which is often referred to as "shuttle" mechanism.
"Just like the two sides of a coin, this unique chemical process enables the battery to work under a wide range of temperatures on the one hand, but induces some problems to the system on the other hand," said Hong-Jie Peng, a graduate student at Department of Chemical Engineering, Tsinghua University. "Soluble polysulfides always flooded to the lithium anode, resulting in parasitic reactions between the lithium anode and redox intermediates. These undesirable reactions drain the battery and become an obstacle to practical applications."
"We are always trying to figure out a more sophisticated strategy to tackle this problem," said Qiang. "When you carefully look into the electrochemical process, you will see something germinated from the old Chinese wisdom, the balance of 'yin' and 'yang'. Generally, every being in this universe can find its counterpart, or the opposite side, and a subtle coexistence of the two keeps a system safe and sound. It gives us a hint about how to mediate the electrochemical reactions in lithium sulfur batteries."

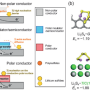
Prof. Zhang and his co-workers offer their new perspective in a recently published paper appearing in the journal Advanced Materials. "We have come up with a design aimed to balance the 'yin' and 'yang' in this system, which are 'sulfiphilicity' and 'lithiophilicity' respectively," Qiang said, adding, "We focus on the culprit, polysulfides, trying to understand how to better trap them and facilitate the conversion. The distinct electronegativity between terminal lithium and sulfur are just like 'yang' and 'yin', the mutual counterpart for each other. Hence, it's a natural guess that an efficient electrochemical mediator that deal with the 'yin' and 'yang' together would be highly efficient."
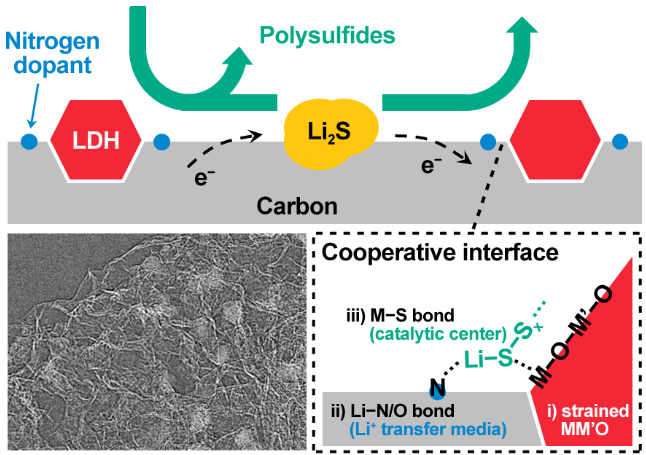
"How to endow one single system with two distinct characters is what we are thinking about, said Zewen Zhang, an undergraduate student in Qiang's group and one of the paper's authors, "Instead of seeking something to intrinsically balance the two, we are doing something more like playing with building bricks. By putting two distinct pieces together, we can fabricate something novel. In our system, we choose layered double hydroxides (LDHs) and nitrogen-doped graphene (NG) to be the building blocks, LDHs for sulfur binding and NG for lithium binding and transferring. The combination of the two is just like the balance of 'yin' and 'yang' in the whole system."
According to their study, this unique design worked out. The combination of the two building blocks offered strong polysulfide adsorption, and enhanced the electrochemical conversion from liquid lithium polysulfides to solid lithium sulfides. "It's very interesting to see that this actually works. Layered double hydroxides possess a self-functional surface of M–O(H) for appropriate chemisorption to polysulfides, while nitrogen-doped graphene provides surface lithium ion binding along with transferring according to lithium bond theory," Zewen said. "Beyond chemisorption, we further find that the cooperation of the two surfaces can facilitate the lithium sulfides nucleation and growth. Then we realized that we have successfully balanced the 'yin' and 'yang' in the lithium sulfur battery, with a subtly designed interface of both 'sulfiphilicity' and 'lithiophilicity'."
These exciting findings drove Qiang's group to fabricate an interlayer with a cooperative interface and to assemble lithium−sulfur batteries with high sulfur loading. Lithium−sulfur batteries with the cooperative interface have demonstrated reined shuttle effect, enhanced capacity, and prolonged cycle life.
"This actually open ups the rational design of advanced materials with cooperative interfaces toward effective regulation of active intermediates," said Qiang. "And we believe that the high-energy-density storage devices with multi-electron chemistries and multi-phase conversion can be further understood through the energy chemistry of cooperative interfaces, or the balance of 'yin' and 'yang' in the system."
"We expect to further extend the concept of cooperative interfaces to other hybrids with two building blocks with unexpected properties for unique applications," Qiang concluded.
More information: Hong-Jie Peng et al. A Cooperative Interface for Highly Efficient Lithium-Sulfur Batteries, Advanced Materials (2016). DOI: 10.1002/adma.201603401
Journal information: Advanced Materials
Provided by Tsinghua University