Driving toward more efficient solar cells

For solar energy to become a real power player in the energy game, solar cells need to be both inexpensive to manufacture and efficient in terms of energy they collect. That's why researchers are focusing their efforts on organic solar cells, which use non-fullerene polymers. These organic polymers are less expensive to produce and some of them, according to some new findings from researchers at NC State and the Hong Kong University of Science and Technology, may have comparable efficiency to their fullerene-based brethren.
NC State physicist Kenan Gundogdu was part of an international team that produced organic solar cells with fast charge separation and low voltage loss.
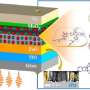
Why is this important? Well, first you have to understand how solar cells work. When sunlight hits a solar cell it is absorbed by the materials in the cell. The light excites these materials, creating a particle called an exciton. The exciton gets split into two charges – an electron (negative) and a hole (positive) – at the interface of two different materials (called donor and acceptor), and then the charges travel to electrodes for collection. Collect enough of these charges, and voila! You can keep the lights on.
However, efficiency is a problem. There are numerous reasons for this, but Gundogdu and colleagues were interested in voltage loss caused by something called the driving force. Since electrons and holes are positively and negatively charged, they attract each other. In organic electronic materials the binding of these particles is so strong that some driving force is necessary to split them. Generally that force comes from the difference in electron and hole attraction preferences of the donor and acceptor materials inside the solar cell. For instance, donor materials will attract holes but not electrons, which leads to separation of the charges at the interface.
Traditionally, a fairly high driving force has been necessary to separate the charges in organic solar cells. The higher the driving force, the faster the charge separation, which sounds like a good thing. But there is no free lunch in physics. With faster charge separation comes more voltage loss, which means the solar cell loses efficiency.
But in new findings that were published online in Nature Energy, the research team created an organic solar cell with both a low driving force and low voltage loss. The solar cell also operated at 9.5 percent efficiency (for comparison, the best fullerene polymer solar cells operate at a bit under 11 percent).
"We are one step closer to realizing inexpensive organic photovoltaic cells with similar performance to traditional commercial solar cells," says Gundogdu. "These findings also improve our understanding of how solar cells work on the molecular level."
More information: Jing Liu et al. Fast charge separation in a non-fullerene organic solar cell with a small driving force, Nature Energy (2016). DOI: 10.1038/nenergy.2016.89
Journal information: Nature Energy
Provided by North Carolina State University