Molecular biology mystery unravelled
The nature of the machinery responsible for the entry of proteins into cell membranes has been unravelled by scientists, who hope the breakthrough could ultimately be exploited for the design of new anti-bacterial drugs. Groups of researchers from the University of Bristol and the European Molecular Biology Laboratory (EMBL) used new genetic engineering technologies to reconstruct and isolate the cell's protein trafficking machinery.
Its analysis has shed new light on a process which had previously been a mystery for molecular biologists.
The findings, published today in the Proceedings of the National Academy of Sciences (PNAS), could also have applications for synthetic biology - an emerging field of science and technology, for the development of novel membrane proteins with useful activities.
Proteins are the building blocks of all life and are essential for the growth of cells and tissue repair. The proteins in the membrane typically help the cell interact with its environment and conserve energy.
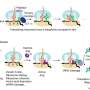
Researchers were able to identify the 'holo-translocon' as the machinery which inserts proteins into the membrane. It is a large membrane protein complex and is uniquely capable of both protein-secretion and membrane-insertion.
Professor Ian Collinson, from the School of Biochemistry at Bristol University said: "These findings are important as they address outstanding questions in one of the central pillars of biology, a process essential in every cell in every organism. Having unravelled how this vital holo-translocon works, we're now in a position to look at its components to see if they can help in the design or screening for new anti-bacterial drugs."
The discovery is a result of an international collaboration between the University of Bristol team and Drs Christiane Schaffitzel and Imre Berger of the European Molecular Biology Laboratory (EMBL) outstation in Grenoble, France.
More information: 'Membrane protein insertion and proton-motiveforce-dependent secretion through the bacterial holo-translocon SecYEG–SecDF–YajC–YidC' by R. Schulze, J. Komar, M. Botte, W. Allen, S. Whitehouse, V. Gold, J. Lycklama a Nijeholt, K. Huard, I. Berger, C. Schaffitzel and I. Collinson in PNAS.
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Bristol