Electrochemical strain microscopy generates new view of fuel cells
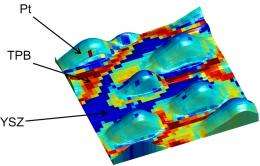
A novel microscopy method at the Department of Energy's Oak Ridge National Laboratory is helping scientists probe the reactions that limit widespread deployment of fuel cell technologies.
ORNL researchers applied a technique called electrochemical strain microscopy that enables them to examine the dynamics of oxygen reduction/evolution reactions in fuel cell materials, which may reveal ways to redesign or cut the costs of the energy devices. The team's findings were published in Nature Chemistry.
"If we can find a way to understand the operation of the fuel cell on the basic elementary level and determine what will make it work in the most optimum fashion, it would create an entirely new window of opportunity for the development of better materials and devices," said co-author Amit Kumar, a research scientist at ORNL's Center for Nanophase Materials Sciences.
Although fuel cells have long been touted as a highly efficient way to convert chemical energy into electrical energy, their high cost -- in large part due to the use of platinum as a catalyst -- has constrained commercial production and consumption.
Large amounts of platinum are used to catalyze the fuel cell's key reaction -- -the oxygen-reduction reaction, which controls the efficiency and longevity of the cell. Yet exactly how and where the reaction takes place had not been probed because existing device-level electrochemical techniques are ill suited to study the reaction at the nanoscale. ORNL co-author Sergei Kalinin explains that certain methods like electron microscopy had failed to capture the dynamics of fuel cell operation because their resolution was effectively too high.
"When you want to understand how a fuel cell works, you are not interested in where single atoms are, you're interested in how they move in nanometer scale volumes," Kalinin said. "The mobile ions in these solids behave almost like a liquid. They don't stay in place. The faster these mobile ions move, the better the material is for a fuel cell application. Electrochemical strain microscopy is able to image this ion mobility."
Other electrochemical techniques are unable to study oxygen-reduction reactions because they are limited to resolutions of 10's of microns – 10,000 times larger than a nanometer.
"If the reaction is controlled by microstructure features that are much finer than a micron, let's say grain boundaries or single extended defects that are affecting the reaction, then you will never be able to catch what is giving rise to reduced or enhanced functionality of the fuel cell," said ORNL's Stephen Jesse, builder of the ESM microscope. "You would like to do this probing on a scale where you can identify each of these defects and correlate the functionality of the cell with these defects."
Although this study mainly focuses on the introduction of a technique, researchers explain their approach as a much-needed bridge between a theoretical and applied understanding of fuel cells.
"There is a huge gap between fundamental science and applied science for energy-related devices like fuel cells and batteries," Kalinin said. "The semiconducting industry, for example, is developing exponentially because the link between application and basic science is very well established. This is not the case in energy systems. They are usually much more complicated than semiconductors and therefore a lot of development is driven by trial and error type of work."
Provided by Oak Ridge National Laboratory

















