Georgia Tech Takes Comprehensive Biofuels Approach

We feel it at the pump. Fuel prices are at record highs and so is the demand for alternative fuels. But major scientific and technological advances are still required before economically viable alternative fuels become a significant part of the U.S. energy supply.
Researchers across the Georgia Institute of Technology campus are focusing their attention on biofuels. And while most experts agree that biofuels are not the silver bullet to solve the world’s long-term fuel needs, they see biofuels as a necessary complement to conventional oil and gas.
Biofuel research at Georgia Tech intensified in 2004 with the launch of the Strategic Energy Institute (SEI), created to enable, facilitate and coordinate programs related to energy research and education.
“Many energy issues are truly multi-disciplinary and can’t be addressed by one faculty member,” says Roger Webb, interim director of the SEI. “The Strategic Energy Institute has been broadly engaging companies to define projects that many faculty members at Georgia Tech can pursue in a collaborative effort.”
This interdisciplinary approach was a major reason why Chevron Corporation chose Georgia Tech as its first strategic research alliance partner, according to Rick Zalesky, vice president of the biofuels and hydrogen unit of Chevron Technology Ventures.
“Georgia Tech has the infrastructure so that researchers from various departments work together in the same building to solve complex problems, and we think that’s terrific,” says Zalesky.
With funding from Chevron, Atlanta startup C2 Biofuels, the Georgia Research Alliance and one of the U.S. Department of Energy’s new BioEnergy Research Centers, Georgia Tech researchers are exploring advanced technologies aimed at making transportation fuels from forestry products.
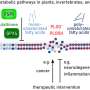
Georgia Tech researchers are examining and optimizing the five major steps required to produce bioethanol, or ethanol obtained from the carbohydrates in many agricultural crops. These steps include selecting the best plant material, preparing the plants for conversion, breaking down the carbohydrates into simple sugars, fermenting the sugars into alcohol and separating the ethanol from water.
Choosing a Plant Source and Preparing It for Conversion
Bioethanol produced from corn is being manufactured at a rate of more than five billion gallons per year in the United States, but concerns exist about the future price and availability of corn as a food crop if it’s being used to help meet energy needs.
Because forest products are a more efficient source of ethanol and more than five million tons of trees are available for harvest each year in Georgia beyond what is needed for pulp mill and sawmill production, Georgia Tech researchers are turning to Southern pine trees.
Switchgrass, a fast-growing tallgrass, is another attractive source of plant material because of its ability to grow in poor soil and adverse climate conditions, its rapid growth and its low fertilization and herbicide requirements.
Art Ragauskas, a professor in the School of Chemistry and Biochemistry, studies the chemistry and structure of the starting plant material, known as biomass, to determine which varieties and characteristics of switchgrass and pine trees improve conversion to ethanol.
He also examines how different acids react with the wood chips to make accessible the complex interior mixture of carbohydrate polymers, including cellulose, hemicellulose and lignin.
“Pre-treatment is performed under severe chemical conditions and very high temperatures. Understanding the chemistry should allow us to make pre-treatments more efficient, less costly and more effective,” says Ragauskas.
After the acid pre-treatment, the wood is placed in a reactor and exposed to high-pressure steam.
John Muzzy, a professor in the School of Chemical and Biomolecular Engineering, and Kristina Knutson, a postdoctoral fellow in the School of Chemistry and Biochemistry, are working with Ragauskas to develop a continuous reactor that will employ mechanical energy and/or boiling water instead of acid and high temperatures to break up the wood. That would greatly reduce processing and chemical costs while increasing the life expectancy of the reactors, Ragauskas notes.
Breaking Down the Sugars and Converting Them to Ethanol
After the pre-treatment, the cellulose and hemicellulose are further broken down to free the sugar for fermentation to alcohol. Commercially available enzymes can do this, but they are too expensive to use in biofuel production, according to Andreas (Andy) Bommarius, a professor in the School of Chemical and Biomolecular Engineering and the School of Chemistry and Biochemistry. As an alternative, he is identifying novel enzymes and engineering them to be longer-lasting and more effective at breaking down cellulose polymers to sugars than those commercially available.
“We want to produce enzymes more efficiently and make them more active and stable, at the same time improving bioethanol production at a lower cost,” explains Bommarius.
In conventional ethanol production, the sugars obtained are then fermented with yeast to produce alcohol. Rachel Ruizhen Chen, an associate professor in the School of Chemical and Biomolecular Engineering, is working to increase the ethanol production rate by using the bacteria Zymomonas mobilis instead of yeast in the fermentation process because it has a three- to five-fold higher productivity than yeast when making bioethanol. Chen plans to manipulate the enzymatic, transport and regulatory functions of the bacterial cell to improve the bioethanol fermentation process.
The lignin portion of the biomass must be extracted from the mixture prior to fermentation. Unfortunately, current pre-treatments break down some of the lignin, which enables it to be carried over to the fermentation process where it acts as a fermentation inhibitor.
William Koros, the Roberto C. Goizueta Chair in the School of Chemical and Biomolecular Engineering, is investigating efficient ways to separate the lignin from the cellulose and hemicellulose portions of the biomass. Koros, a Georgia Research Alliance (GRA) eminent scholar in membranes, plans to extract the lignin byproducts by pulling the hydrolyzed biomass mixture through a selective membrane with a vacuum using a process called pervaporation.
Lignin is an important by-product of the enzymatic process and has many potential uses. Ragauskas is examining the possibility of converting lignin to a biofuel precursor or using lignin as a building block chemical to make new polymers or chemicals. Professors Christopher Jones and Pradeep Agrawal, both of the School of Chemical and Biomolecular Engineering, are exploring ways to chemically fractionate pine and convert suitable portions to true gasoline fuels.
To produce a biofuel with a similar energy density to gasoline from renewable feedstocks, they plan to convert pre-treated pine to fuel using chemical catalysts traditionally used by the petroleum industry, rather than enzymes. These biofuels could yield higher miles-per-gallon than traditional ethanol-rich fuels such as E-85, according to Jones.
Separating Ethanol from Water
For bioethanol, once the sugars are fermented into alcohol, a significant amount of water must be separated out. This separation primarily occurs in a distillation column, which involves heating the mixture and separating the components by the differences in their boiling points.
“Distillation is very energy intensive and expensive, and it might defeat the purpose when you’re trying to produce biofuel economically,” says Sankar Nair, an assistant professor in the School of Chemical and Biomolecular Engineering, who is collaborating with Koros on two separation projects aimed at improving the energy efficiency of the biofuel process.
A membrane-based approach would avoid the need to supply heat energy, and instead rely on differences in the transport rates of the components through a membrane to achieve separation. The challenge is in producing selective membrane systems that can produce pure ethanol. Polymer materials have been widely investigated and have the advantage of high throughput, but such membranes can’t yet produce pure ethanol from a dilute ethanol-water mixture, notes Nair.
Instead, Koros and Nair are exploring membranes that contain nanoparticles of porous inorganic materials called zeolites that are so small they can be dispersed efficiently into a polymer matrix. The very specific porosity of the zeolite should allow separation of ethanol from water. By using two membranes in series – the first hydrophobic to remove ethanol from a large mass of water and the second hydrophilic to remove any trace water in the ethanol product from the first membrane – it may be possible to design an economical membrane process for biofuel separation from water.
Taking a Systems Approach
Producing ethanol from biomass involves more than these process steps. Researchers must also decide how to ship the biomass to the processing plant, how large the processing plant should be, where it should be located, and how to ship the ethanol to fueling stations.
Bill Bulpitt, an SEI senior research engineer who returned to Georgia Tech in 2004 after working 17 years for Southern Company, is working with students who are running computer simulation models that represent what a full-scale production plant might look like. The models analyze the costs for the various components of the system, which helps to determine the optimal biorefinery size.
“When building a biorefinery, there is a certain size that’s economically viable. That’s what we are trying to determine,” Bulpitt explains.
To evaluate a biofuel system, the project team must consider the energy balance – that is, how much energy goes in versus how much comes out. A biofuel system must take into account positive or negative energy balances, positive or negative net greenhouse gas emissions, and positive or negative environmental and ecosystem impacts.
Ethanol biorefineries could get a significant economic boost from the sale of high-value chemicals that could be generated from the same feedstock. Charles Eckert, a professor in the School of Chemical and Biomolecular Engineering and collaborators Charles Liotta and Art Ragauskas are exploring the use of environmentally friendly solvent and separation systems to produce specialty chemicals, pharmaceutical precursors and flavorings from a small portion of the ethanol feedstock.
Matthew Realff, an associate professor in the School of Chemical and Biomolecular Engineering, is developing optimization models to determine the best structure for a biofuel processing system. Realff’s model integrates information from crop production through processing to fuel distribution. It includes information on the location and number of crop acres available, the current economic value of the crop, distances and ability to ship the crop, the economic scaling of the cost of the processing equipment with size and the location of the distribution terminals.
These optimization models are valuable to companies like C2 Biofuels that plan to build biorefineries. And they complete the comprehensive research approach Georgia Tech has taken toward optimizing bioethanol production process.
“Researchers at Georgia Tech have different strengths and take different approaches toward solving the problem of developing biofuels,” says Christopher Jones. “If you assemble all of the pieces together, you will come up with the best solution.”
This article originally appeared in the Summer 2007 issue of Research Horizons, Georgia Tech’s research magazine.
Source: Georgia Institute of Technology