Fast and efficient biologically inspired catalyst could someday make fuel cells cheaper
To make fuel cells more economical, engineers want a fast and efficient iron-based molecule that splits hydrogen gas to make electricity. Online Feb. 17 at Nature Chemistry, researchers report such a catalyst. It is the first iron-based catalyst that converts hydrogen directly to electricity. The result moves chemists and engineers one step closer to widely affordable fuel cells.
"A drawback with today's fuel cells is that the platinum they use is more than a thousand times more expensive than iron," said chemist R. Morris Bullock, who leads the research at the Department of Energy's Pacific Northwest National Laboratory.
His team at the Center for Molecular Electrocatalysis has been developing catalysts that use cheaper metals such as nickel and iron. The one they report here can split hydrogen as fast as two molecules per second with an efficiency approaching those of commercial catalysts. The center is one of 46 Energy Frontier Research Centers established by the DOE Office of Science across the nation in 2009 to accelerate basic research in energy.
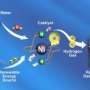
Fuel cells generate electricity out of a chemical fuel, usually hydrogen. The bond within a hydrogen molecule stores electricity, where two electrons connect two hydrogen atoms like a barbell.
Fuel cells use a platinum catalyst—essentially a chunk of metal—to crack a hydrogen molecule open like an egg: The electron whites run out and form a current that is electricity. Because platinum's chemical nature gives it the ability to do this, chemists can't simply replace the expensive metal with the cheaper iron or nickel. However, a molecule that exists in nature called a hydrogenase (high-dra-jin-ace) uses iron to split hydrogen.
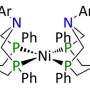
Bullock and his PNNL colleagues, chemists Tianbiao "Leo" Liu and Dan DuBois, have taken inspiration for their iron-wielding catalyst from a hydrogenase. First Liu created several potential molecules for the team to test. Then, with the best-working molecule up to that point, they determined and tweaked the shape and the internal electronic forces to make additional improvements.
One of the tricks they needed the catalyst to do was to split hydrogen atoms into all of their parts. If a hydrogen atom is an egg, the positively charged proton that serves as the nucleus of the atom would be the yolk. And the electron, which orbits around the proton in a cloud, would be the white. The catalyst moves both the proton-yolks and electron-whites around in a controlled series of steps, sending the protons in one direction and the electrons to an electrode, where the electricity can be used to power things.
To do this, they need to split hydrogen molecules unevenly in an early step of the process. One hydrogen molecule is made up of two protons and two electrons, but the team needed the catalyst to tug away one proton first and send it away, where it is caught by a kind of molecule called a proton acceptor. In a real fuel cell, the acceptor would be oxygen.
Once the first proton with its electron-wooing force is gone, the electrode easily plucks off the first electron. Then another proton and electron are similarly removed, with both of the electrons being shuttled off to the electrode.
The team determined the shape and size of the catalyst and also tested different proton acceptors. With the iron in the middle, arms hanging like pendants around the edges draw out the protons. The best acceptors stole these drawn-off protons away quickly.
With their design down, the team measured how fast the catalyst split molecular hydrogen. It peaked at about two molecules per second, thousands of times faster than the closest, non-electricity making iron-based competitor. In addition, they determined its overpotential, which is a measure of how efficient the catalyst is. Coming in at 160 to 220 millivolts, the catalyst revealed itself to be similar in efficiency to most commercially available catalysts.
Now the team is figuring out the slow steps so they can make them faster, as well as determining the best conditions under which this catalyst performs.
More information: Nature Chemistry 2013, doi:10.1038/NCHEM.1571
Journal information: Nature Chemistry
Provided by Pacific Northwest National Laboratory