New metal catalyst drives hydrogen fuel reaction forwards and backwards
(PhysOrg.com) -- When it comes to driving hydrogen production, a new catalyst built at Pacific Northwest National Laboratory can do what was previously shown to happen only in nature: store energy in hydrogen and release that energy on demand. This new nickel-based complex drives the reaction but is not consumed by it. While slow, the catalyst wastes little energy. It turns electrons and protons into hydrogen. The hydrogen molecule holds the energy in a very small space until it is needed. The same catalyst then breaks the single bond in the hydrogen molecule, releasing electrons to do work.
Reducing our reliance on fossil fuels benefits the economy, national security, and the environment. However, solar and wind power cannot be major players on the energy stage until the intermittent power they generate can be stored and used when needed. One option is to transform the electrical energy from solar and wind into hydrogen, which can be used in fuel cells. To create the hydrogen, scientists want a single, efficient catalyst, which had eluded them. This research proves that such a catalyst can be synthesized.
"We are trying to build metal catalysts that will convert between electrical and chemical energy to make it possible to use renewable sources," said Dr. Morris Bullock, who worked on the research at PNNL and is the Director of the Center for Molecular Electrocatalysis.
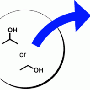
Often learned in high school chemistry classes, the reaction for working with hydrogen looks pretty simple:

"However, the mechanism is remarkably complicated," said Bullock. "There is a lot of detail in this process: taking the hydrogen apart, moving protons and electrons, and putting it back together."
The team began with the type of catalyst they've worked with for more than two years at the Center for Molecular Electrocatalysis. The catalyst relies on a nickel center or active site to do the work. This metal was chosen for its low cost and abundance.
"Replacing fossil fuels with devices that require precious metals is simply not reasonable," said Bullock.
Wrapped around and attached to the nickel active site are several molecular strands or ligands. These ligands function as arms, transporting molecules, protons, and electrons to and from the active site. The team systematically explored how changing the size, structure, and behavior of the ligands affected the reaction. They characterized each version of the catalyst using nuclear magnetic resonance spectroscopy and electrochemical measurements.
With the catalyst characterized, they tested its ability to drive the reaction forward and back. The tests involved measuring the electric current produced by adding hydrogen to the catalyst. Using complex mathematical formulas, they determined the speed and efficacy of the reactions.
The catalyst proved very efficient, wasting little energy. Energy waste is measured by determining the overpotential, a ratio of energy used under real world conditions versus the energy needed under perfect conditions. "This [catalyst] has a lower overpotential than we usually find," said Bullock. "Sadly, it is also slow."
Speed. The team is working to speed up the catalyst by tweaking the molecular structure of the ligands to transport protons to and from the active site more quickly.
"We'll figure out what the slow step is and then figure out how to speed it up. Then, we'll take on the next slowest step, and so on, until we get the speed we need," said Bullock.
More information: SE Smith, JY Yang, DL DuBois, and RM Bullock. 2012. "Reversible Electrocatalytic Production and Oxidation of Hydrogen at Low Overpotentials by a Functional Hydrogenase Mimic." Angewandte Chemie. DOI: 10.1002/anie.201108461
Journal information: Angewandte Chemie
Provided by Pacific Northwest National Laboratory