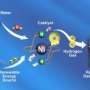
Iron center and pendant nitrogen needed to achieve catalyst's goal
(Phys.org) -- To crack hydrogen molecules and free the electrons, scientists at Pacific Northwest National Laboratory built nature-inspired molecules that get the job done. These designer molecules, or catalysts, rely on an iron center and small, dangling molecular chains with strategically placed nitrogen atoms. Known as pendant amines, these chains draw in the hydrogen molecule and position it just so. The iron center breaks apart the hydrogen into protons and electrons. The pendant amines shuttle the protons off, and the process starts all over again.
"We're not trying to precisely mimic nature, just incorporate the salient features that make natural catalysts function," said Dr. Morris Bullock, Director of the Center for Molecular Electrocatalysis, led by PNNL. The results were published in the Journal of the American Chemical Society.
Using fossil fuels to power cars and heat homes continues to raise economic, environmental, and security issues. A popular alternative is the fuel cell, which converts hydrogen, methanol, or other chemicals into water, and in the process it turns out electricity. But the catalyst of choice for the fuel cells is platinum, which is expensive and scarce. In contrast, iron is far less expensive and is the Earth's most abundant metal.
The PNNL team based their design on natural hydrogenase enzymes, fast and energy-efficient molecular catalysts that can split the hydrogen molecules and put the electrons and protons back together to create hydrogen. Their active sites contain the inexpensive and abundant metals iron and/or nickel, which makes them attractive to scientists who want to develop a cheaper, easily available catalyst mimicking the structure and function of hydrogenases.
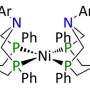
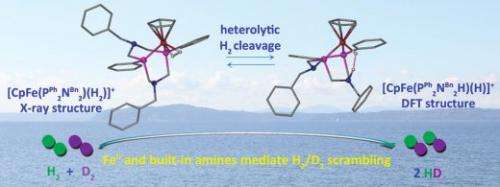
Dr. Tianbiao (Leo) Liu, a postdoctoral fellow at PNNL, led the catalyst design. He and his colleagues synthesized and characterized a series of iron complexes using NMR spectroscopy, electrochemical studies, and x-ray diffraction. In these complexes, the pendant amines are a critical feature. They facilitate the coupling of proton and electron transfer reactions and proton transfer between acids and bases in solution and the active iron center.
Liu incorporated an amine (nitrogen-containing) pendant into a larger phosphorus-containing, six-sided ring, called a phosphine ligand, at just the right place to where it "hangs around" the ligand in a configuration that is structurally close enough to interact with the hydrogen.
"Previous research has established that pendant amines can play an important role in relaying protons between metal and solution, in coupling proton and electron transfers, and in activating dihydrogen," said Liu. "We confirmed that both the iron center and pendant amines are crucial to hydrogen bonding and cleavage."
As a control experiment, the scientists also made a closely related compound without pendant amines, which didn't react nearly as fast. It confirmed that the pendant amine is required to make the cleaving happen.
They showed that the compound will break the H-H bond into two ions, with the proton (H+) landing on the nitrogen, and the hydride (H-) being transferred to the iron. Unambiguous proof came from using nuclear magnetic resonance spectroscopy. They took the iron catalyst with a one-to-one mixture of hydrogen and deuterium. Deuterium is just a tiny bit different from hydrogen; this difference makes it easy to track in the spectrometer. Both H-H and D-D bonds broke, making HD, proving that the bond had cleaved.
This cleavage occurred at room temperature under mild conditions, thus it mimics conditions of typical low-temperature fuel cells.
The scientists have done the hardest part by creating the first iron complexes that will cleave H2 in just the right way. That will provide most of the steps needed-but not quite.
"Catalytic oxidation of H2 releases electrons. But in the process, a bunch of protons are also made. So we added a base to take up the protons," said Bullock. "The base binds to the metal of our iron catalysts, so they are not an electrocatalyst for complete oxidation of H2. That was a bit of a surprise to us, but a valuable one." He added, "We have to make sure we understand the energies of all these compounds so we can match the ability of a pendant amine to accept protons with the ability of iron to accept hydride."
More information: TL Liu, et al. 2012. "Synthesis, Characterization and Reactivity of Fe Complexes Containing Cyclic Diazadiphosphine Ligands: The Role of the Pendant Base in Heterolytic Cleavage of H2." Journal of the American Chemical Society 134(14):6257-6272. DOI:10.1021/ja211193j
Journal information: Journal of the American Chemical Society
Provided by Pacific Northwest National Laboratory