This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Chemistry inspired by one-pot cooking helps achieve optimal reaction conditions
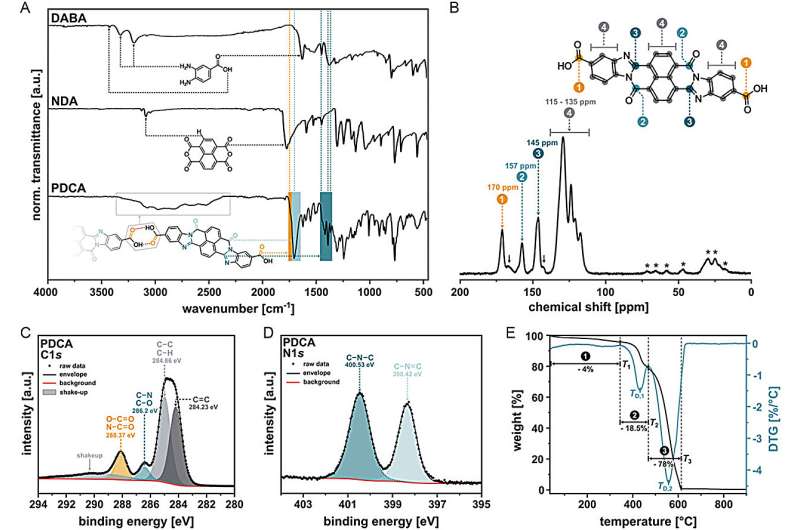
People have long dreamed of developing materials to circumvent the challenges of daily life. Ideally, one could capitalize on a combination of the features of different materials, profiting from their advantages while avoiding the disadvantages. In chemistry, this concept has been applied to hybrid materials, particularly with the combination of organic and inorganic compounds.
Organic materials are known for their functional diversity, while inorganic materials offer superior stability. However, the fusion of organic and inorganic substances poses significant challenges due to the differing reaction conditions required for their formation. The research team led by Professor Miriam Unterlass at the University of Konstanz employs a method that balances these contrasting conditions, allowing the reactions to occur simultaneously and synergistically in a single reaction vessel. Chemists have named this method "one-pot synthesis."
The researchers have fine-tuned this method to achieve optimal reaction conditions, akin to cooking a perfect one-pot meal, where each ingredient must be prepared just to the right state. Key to their success is the precise control of pressure, temperature, and time, along with the right selection of available ingredients, or as chemists call them, starting materials.
"The beauty of our approach lies in its simplicity," says Frank Sailer, who played a significant role in one-pot synthesis while pursuing his doctoral thesis. "We avoid toxic catalysts and solvents, using only isopropanol, a common disinfectant, as a solvent. This makes our process both sustainable and environmentally friendly."
The resulting materials, named pigment@TiO2 are composed of special colorant molecules, known as organic pigments, and layered titanium dioxide. Are the results of one-pot synthesis truly the best of both worlds? Actually, they are much more than that.
Sailer says, "We don't just want to achieve a sum of all the properties of both individual components but new, synergetic properties that none of the individual materials exhibits." These synergistic properties make pigment@TiO2 materials particularly suitable for battery applications.
Why was this approach not employed to make such hybrid materials in the past? "This is because the idea is very unusual. The organic components are typically not synthesized under the employed reaction conditions," Sailer explains.
Finding such materials and reaction pathways is one of the research goals pursued by Unterlass' research team. They study how chemical synthesis processes can be optimized to lead to better, more sustainable advanced materials. "We aim to produce better materials faster and in a more environmentally friendly way," says Unterlass.
The detailed findings of Unterlass' research team are published in the journal Small Structures.
More information: Frank Sailer et al, Covalently Linked Pigment@TiO2 Hybrid Materials by One‐Pot Solvothermal Synthesis, Small Structures (2024). DOI: 10.1002/sstr.202400074
Provided by University of Konstanz



















