This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Ion swap dramatically improves performance of CO₂-defeating catalyst
A team of scientists led by the Department of Energy's Oak Ridge National Laboratory has found an unconventional way to improve catalysts made of more than one material. The solution demonstrates a path to designing catalysts with greater activity, selectivity and stability.
A catalyst normally uses a support to stabilize nanometer-sized metal particles that speed important chemical reactions. The support, through interactions with the metal particles, also helps create a unique interface with sites that can dramatically enhance reaction rate and selectivity. To improve catalytic efficiency, researchers typically try different combinations of metals and supports. ORNL's team instead focused on implanting specific elements right next to metal nanoparticles at their interface with the support to boost catalytic efficiency.
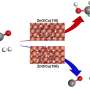
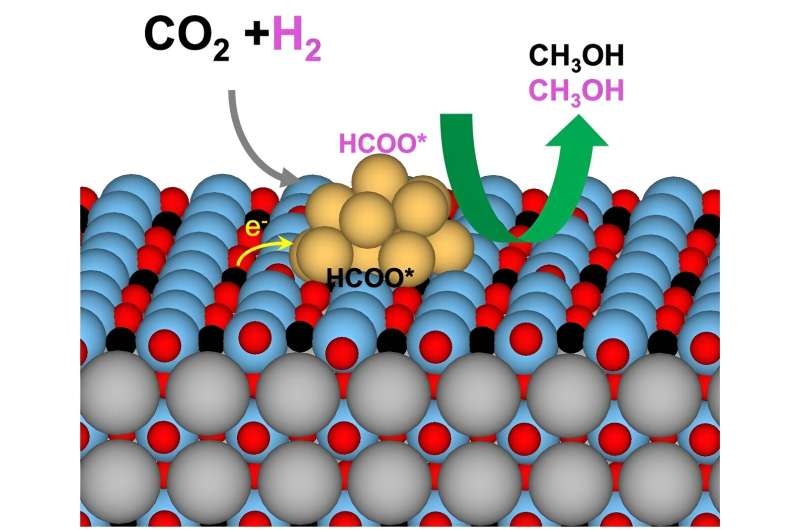
The researchers studied a catalyst that hydrogenates carbon dioxide to make methanol. Its copper nanoparticles are supported by barium titanate. In the crystalline support, two positively charged ions, or cations, pair with negatively charged ions, or anions. When the team extracted partial oxygen anions from the support and implanted hydrogen anions, this ion swap altered the reaction kinetics and mechanisms and resulted in triple the yield of methanol.
"Tuning the anion site of the catalyst support can greatly impact the metal-support interface, which leads to enhanced conversion of waste carbon dioxide to valuable fuels and other chemicals," said project head Zili Wu, leader of ORNL's Surface Chemistry and Catalysis group.
The research, published in Angewandte Chemie International Edition, is featured on the journal's back cover. The findings point to a unique role that hydrogen anions, or hydrides, could play in boosting the performance of catalysts that turn carbon dioxide into methanol. Wu's team was the first to use anion substitution to this end. Such catalysts could join the portfolio of technologies aimed at achieving global net-zero carbon dioxide emissions by 2050.
In designing the catalyst, the team chose the perovskite barium titanate for the support. It is one of the few materials in which hydrogen anions, which are highly reactive to air or water, can be incorporated to form a stable oxyhydride. Moreover, the scientists hypothesized that the incorporated hydrogen anions might affect the electronic properties of neighboring copper atoms and participate in the hydrogenation reaction.
"A perovskite allows you to tune not only the cations almost across the periodic table, but also the anion sites," said Wu. "You have a lot of tuning 'knobs' to understand its structure and catalytic performance."
The hydrogenation of carbon dioxide to make methanol requires high pressure—more than several tens of times the pressure of Earth's atmosphere at sea level. Probing the catalyst under resting ("in situ") versus working ("operando") conditions took expertise and equipment that are hard to find outside national labs. This reaction has been studied for decades, but its active catalytic sites and mechanisms had remained unclear until now because of the dearth of in situ/operando studies.
"I'm really proud that we pulled from diverse teams to illuminate the underlying mechanism," Wu said.
"We combined multiple in situ and operando techniques to characterize the structure of copper, the support and the interface under reaction conditions," said ORNL co-author Yuanyuan Li. She uses spectroscopy to reveal the dynamic atomic, chemical and electronic structure of materials under synthesis and reaction conditions. "Copper can quickly change after being exposed to the air or other environments. It was therefore very important for us to reveal the structure of the catalyst under real working conditions and then correlate that with its performance."
To reveal the structure of the catalyst under working conditions, Li and former ORNL postdoctoral fellow Yang He went to the Stanford Synchrotron Radiation Lightsource at SLAC National Accelerator Laboratory. With SLAC's Jorge Perez-Aguilar in the laboratory of Simon Bare, they used in situ X-ray absorption spectroscopy to reveal the structure of the copper nanoparticles under high-pressure reaction conditions. The researchers collaborated through the Consortium for Operando and Advanced Catalyst Characterization via Electronic Spectroscopy and Structure, or Co-ACCESS.
Back at ORNL's Center for Nanophase Materials Sciences, a DOE Office of Science user facility, ORNL Corporate Fellow Miaofang Chi and ORNL postdoctoral fellow Hwangsun "Sunny" Kim performed scanning transmission electron microscopy to compare the copper structure before and after the chemical reaction.
Moreover, ORNL staff scientists Luke Daemen and Yongqiang Cheng performed in situ high-pressure inelastic neutron scattering at the VISION beamline of the Spallation Neutron Source, a DOE Office of Science user facility, to characterize the structure of the hydride in the oxyhydride support. Because neutrons are sensitive to lightweight elements, they were used to monitor the hydride structure after reaction at high pressures. It remained stable.
At Vanderbilt University, postdoctoral fellow Ming Lei with Professor De-en Jiang used density functional theory to calculate electronic structure of the material. The theory-based calculations and experimental results together showed that hydrides on the support directly participated in hydrogenating carbon dioxide to make methanol and altered the electronic state of copper to enhance methanol-producing reactions at the interface.
To learn more about the kinetics and mechanism of the chemical reaction, He, with ORNL staff member Felipe Polo-Garzon, customized a technique called steady-state isotopic transient kinetic analysis, or SSITKA, for use under high-pressure conditions. They coupled it with an operando high-pressure technique called diffuse reflectance infrared spectroscopy, or DRIFTS.
"We developed the method under real reaction conditions to understand both reaction kinetics and mechanisms," said He, now with DOE's Pacific Northwest National Laboratory. "That's going to contribute to the field by filling the gap between ambient and higher-pressure studies."
SSITKA suggested that the hydride-rich perovskite had a higher density of sites that were more active and selective for methanol production. The addition of DRIFTS revealed that a chemical species called formate—carbon dioxide with a hydrogen atom connected—was the major reaction intermediate. DRIFTS-SSITKA also showed that subsequent steps to hydrogenate formate into methanol limit the rate of the reaction.
Next, Wu and colleagues will change the reactivity of the hydride in the support by changing the perovskite's composition.
"Then you potentially can further increase the performance of your catalyst," Wu said. "This approach of anion tuning of catalysts provides a new paradigm for controlling chemical reactions."
More information: Yang He et al, Significant Roles of Surface Hydrides in Enhancing the Performance of Cu/BaTiO2.8H0.2 Catalyst for CO2 Hydrogenation to Methanol, Angewandte Chemie International Edition (2023). DOI: 10.1002/anie.202313389
Journal information: Angewandte Chemie International Edition
Provided by Oak Ridge National Laboratory