This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study shows 3D-covalent organic frameworks can be tuned using structural isomers
Porous solids are abundant in the world. Examples include activated carbon, zeolite, and metal-organic frameworks (MOFs). Solid electrolytes in next-generation batteries have ion migration channels and hence are porous solids in a broad sense. Because the properties of porous solids are dictated by pore size, the way the pores are connected internally, and the chemical nature of the pore walls, achieving high degrees of freedom in designing these properties is crucial.
As a novel class of nanoporous solids, covalent organic frameworks (COFs) are formed by covalently and repeatedly condensing building block molecules. COFs have recently been extensively studied because they offer high design freedom in terms of function and microscopic geometries in the materials, as well as the high thermal stability that is desired for applications.
Three-dimensional COFs (3D-COFs), as a subclass of COFs, are expected to be useful due to their inherently richer framework topologies than that of preceding two-dimensional COFs; most of the previous COF studies were for 2D-COFs.
However, there has been a dilemma. COFs have more stable and harder covalent bonds than MOFs, which have weaker and softer coordination bonds. This results in an advantage and two disadvantages. An advantage is the higher stability that enhances the durability during use. The first disadvantage is the poorer topological diversity in the framework geometries so far achieved. The second disadvantage is the difficulty in obtaining COFs with high crystallinity to the extent that crystal shapes are recognizable using an optical microscope.
Both these disadvantages arise from the same root—the highly stiff and directional nature of covalent bonds (as compared to less stiff and less directional coordination bonds constituting MOFs). Addressing these disadvantages has been inevitable to boost applications of 3D-COFs.
To resolve these issues, a research team led by Professor Yoichi Murakami of the Laboratory for Zero-Carbon Energy at the Institute of Innovative Research, Tokyo Institute of Technology attempted to create a new type of 3D-COFs by combining building block molecules that contain flexible moieties. The study is published in the Journal of the American Chemical Society.
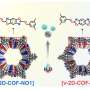
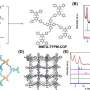
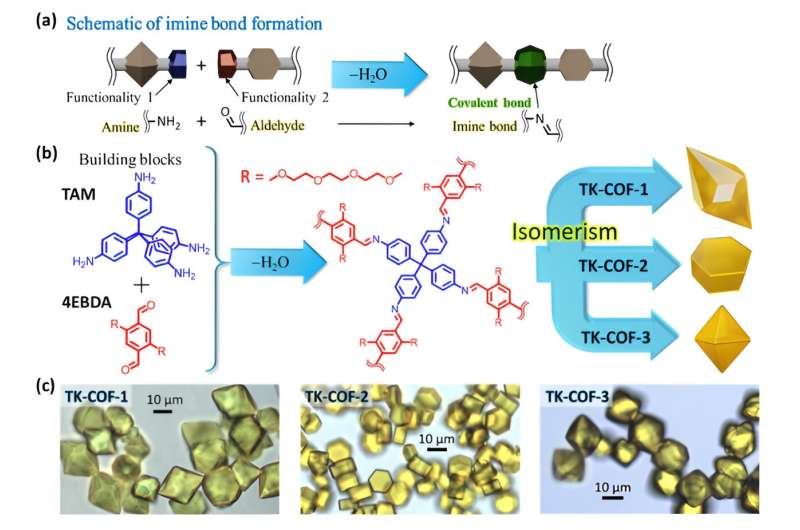
As shown by Figures 1a and b, they selected TAM, which is a monomer with angular flexibility in the directions of the four amine functionalities, and 4EBDA, which is a monomer with two flexible side chains and two aldehyde functionalities, to condense them by forming imine bonds—one of the well-known covalent bonds in organic compounds.
The sidechain ("R" in Figure 1b) is a part of polyethylene glycol, which is a functionality known to enhance the transport of lithium ions and is therefore usable as a solid-state electrolyte in lithium-ion batteries.
Significantly, after repeated trials of many formation conditions in solution, the research group produced beautiful, high-quality crystals with three distinctly different shapes shown in Figure 1c. The researchers named these crystals TK-COF-1, TK-COF-2, and TK-COF-3. Surprisingly, the research team discovered that these three new COFs had an identical chemical composition.
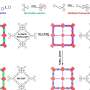
The X-ray diffraction measurements of the crystals revealed their qualitatively different nanostructures as shown in Figures 2a to c. The framework topologies of these crystals were found to be dia (TK-COF-1), qtz (TK-COF-2), and dia-c3 (TK-COF-3).
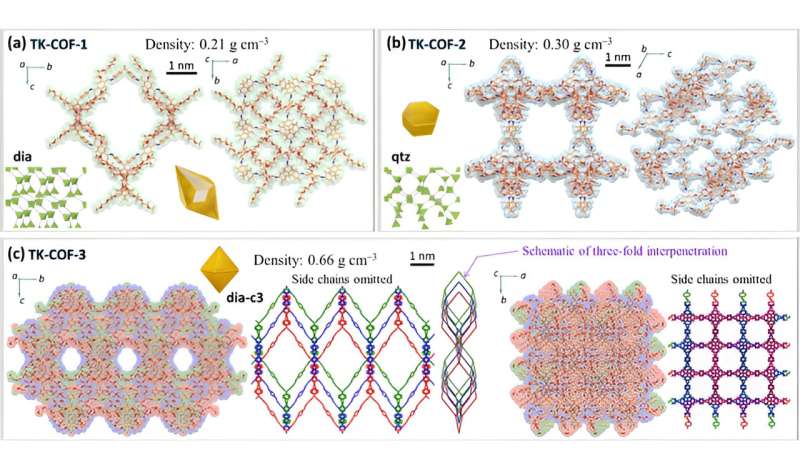
This is an important finding that the diversity of structures and properties of 3D-COFs can be increased not only by the conventional way of choosing block molecules but also by controlling the emergence of isomer type during polycondensation of building block molecules. The density of COFs (indicated in the figure panels) was successfully changed by a factor of about 3 due to the choice of isomers, as indicated in the panels of Figure 2.
In summary, this work demonstrates that key properties of 3D-COFs (density, pore size, mechanical properties, etc.) can be tuned with a new degree of freedom utilizing the emergence and control of structural isomerism. This outcome will accelerate the future applications of COFs.
More information: Xiaohan Wang et al, Triple Isomerism in 3D Covalent Organic Frameworks, Journal of the American Chemical Society (2024). DOI: 10.1021/jacs.3c13863
Journal information: Journal of the American Chemical Society
Provided by Tokyo Institute of Technology