This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers discover important membrane transport mechanism in pathogenic bacteria
Some bacterial membrane transporters work almost like freight elevators to transport substances through the cell membrane into the interior of the cell. The transporter itself spans the bacterial membrane. Like a forklift, a soluble protein outside the bacterium transports the substance to the "elevator" and unloads its cargo there. The freight elevator transports it to the inside of the cell, in other words to another floor.
Researchers at the University Hospital Bonn (UKB) and the University of Bonn, in collaboration with a team from the University of York, have now studied the interaction between the transporter and its soluble substrate binding protein. Interestingly, they adapt precisely to each other during the transportation process.
Because this happens very quickly, the researchers virtually "blocked" the elevator by specifically inserting anchors, so-called disulfide bridges. This enabled them to prove that only the loaded "forklift" fits the elevator if it is on the right floor. This makes transportation really effective. The study has now been published in the journal Nature Communications.
Like all cells, bacteria are also surrounded by a cell membrane. This thin layer of lipids encloses nutrients, genetic material and proteins of the cell. However, certain substances must be able to pass through the membrane. For example, substances that help pathogens to evade the human body's immune response.
To this end, pathogenic bacteria such as Haemophilus influenzae have so-called ATP-independent periplasmic (TRAP) transporters. They have two very flexible transmembrane domains that span the cell wall. The pathogen Haemophilus influenzae uses the TRAP transporter to transport sialic acid from its environment into the cell interior, which is then incorporated into the bacterial cell wall.
The small sugar molecule is very common in human tissue. "Once it is incorporated into the bacterial cell wall, the sialic acid acts like a camouflage cap for the bacteria. This allows them to hide from our immune system because it makes them look similar to the body's own tissue," says PD Dr. Gregor Hagelueken from the Institute of Structural Biology at the UKB. He is also a member of the Transdisciplinary Research Area (TRA) "Life & Health" and the Immunosensation2 Cluster of Excellence at the University of Bonn.
Protein catches substance like a Venus flytrap
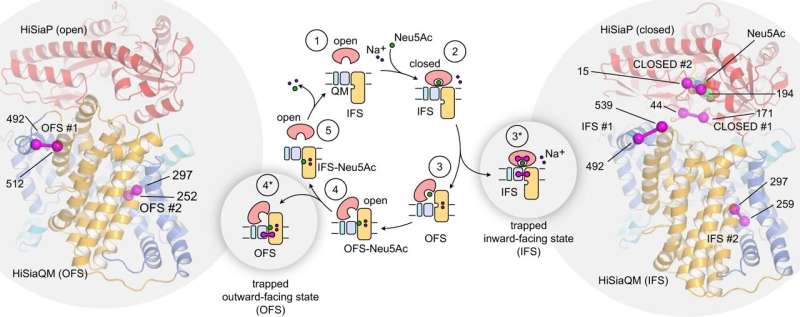
The TRAP transporter is supported by an additional substrate binding protein (SBP), which searches for sialic acid outside the bacterial membrane. Once this "forklift" has found a sugar molecule, the SBP changes its shape and binds the sialic acid tightly. "This binding process can be compared to the snapping shut of a Venus flytrap," says Philipp Hendricks, one of the first authors of the study and a doctoral student at the Institute of Structural Biology at the University of Bonn.
It has long been suspected that the TRAP transporter recognizes the closed form of the substrate binding protein. The Bonn researchers therefore investigated whether the opening and closing of the SBP and the upward and downward movement of the TRAP transporter are actually coupled to each other.
Because these movements are very fast, Hagelueken's team used what is known as disulfide engineering, a special biotechnological tool, to block the transporter. "We locked the 'elevator' on different floors, so to speak—either inside the cell or outside," says first author Dr. Martin Peter. He was a postdoctoral researcher at the Institute of Structural Biology and is now a researcher at Heidelberg University.

Bacterial freight elevator made visible with fluorescence microscopy
In collaboration with the laboratory of Prof. Dr. Ulrich Kubitscheck at the Clausius Institute for Physical and Theoretical Chemistry at the University of Bonn, the researchers were able to incorporate the TRAP transporter into artificial membranes. Using single-molecule fluorescence microscopy, they were able to precisely observe the binding between the TRAP transporter and the "forklift" and thus watch them live at work.
"Our experiments showed that the movements of the SBP and those of the transmembrane elevator are indeed coupled with each other," says first author Jan A. Ruland, postdoctoral researcher at the Clausius Institute at the University of Bonn. Thus, the SBP in its closed state preferentially binds to the inward-facing membrane elevator and the "open" SBP to the outward-facing TRAP transporter.
"These new insights into the mechanism of the transporter may help in the future to develop antibiotics that ensure that the elevators of bacteria get stuck. This would put an end to the game of hide-and-seek and our immune system could destroy the bacteria more easily," says Hagelueken from the UKB.
More information: Martin F. Peter et al, Conformational coupling of the sialic acid TRAP transporter HiSiaQM with its substrate binding protein HiSiaP, Nature Communications (2024). DOI: 10.1038/s41467-023-44327-3
Journal information: Nature Communications
Provided by University of Bonn