This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New technique could make modeling molecules much easier
Much like the humans that created them, computers find physics hard, but quantum mechanics even harder. But a new technique created by three University of Chicago scientists allows computers to simulate certain challenging quantum mechanical effects in complex electronic materials with far less effort.
By making these simulations more accurate and efficient, the scientists hope the technique could help discover new molecules and materials, such as new types of solar cells or quantum computers.
"This advance holds immense potential for furthering our understanding of molecular phenomena, with significant implications for chemistry, material science, and related fields," said scientist Daniel Gibney, a University of Chicago Ph.D. student in chemistry and first author on the paper, published Dec. 14 in Physical Review Letters.
Electrons and energy
A leaf or a solar panel looks smooth and simple from the outside, but zoom down to the molecular level and you'll see a wildly complicated dance of electrons and molecules.
In order to make new advances in sustainability, manufacturing, agriculture, and many other fields, scientists model the behavior of these chemical and molecular interactions. This helps reveal new design possibilities for the future—for everything from new ways to sequester carbon dioxide to new types of quantum bits.
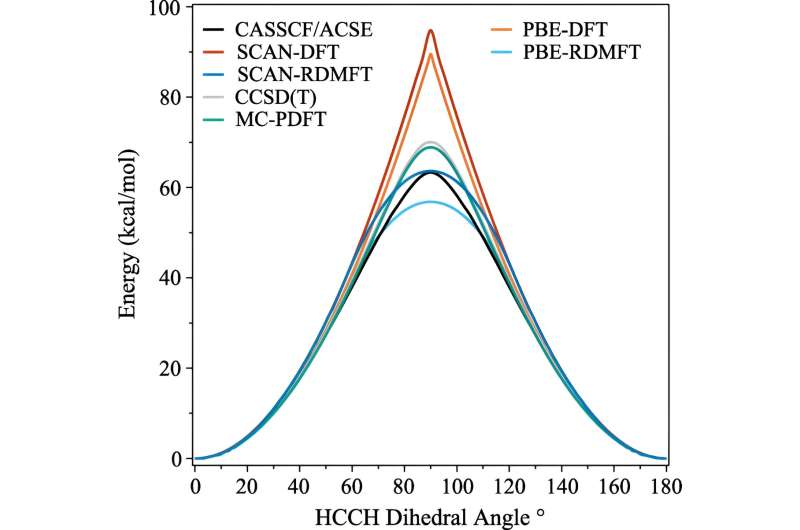
Many strides have been made in the last decades, but one of the areas that has remained stubbornly difficult to simulate is when the molecules start to display complex quantum mechanical behaviors that scientists call strong correlation.
The trouble is that once electrons start to flaunt their most quantum-mechanical effects—such as becoming "entangled"—the calculations instantly need much more computing power. Even supercomputers struggle to handle the implications.
One of the commonly used calculations is called density functional theory. "This is basically the most omnipresent technique for predicting electronic structure, but it's essentially an approximation where all the electrons are treated as a function of one electron," explained David Mazziotti, professor of chemistry and senior author on the study.
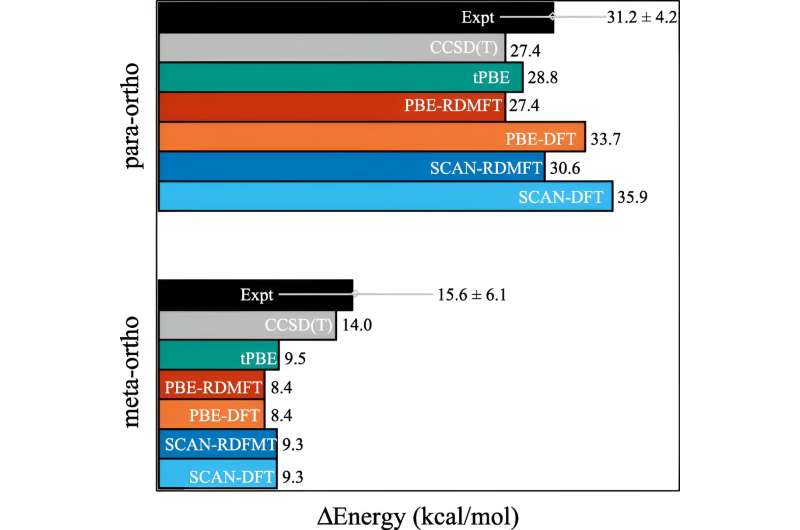
For many calculations, an approximation does the job. But it starts to break down as the behavior of the electrons becomes more correlated, as happens when quantum mechanics starts to come into play. In quantum mechanics, these electrons can be in multiple places, or orbitals, simultaneously. This stymies not only human brains, but also density functional theory.
"And this is an important problem, because a lot of the issues we care about in the 21st century—like new molecules and materials for renewable energy and sustainability—require us to exploit the quantum nature of materials," said Mazziotti.
Mazziotti, Gibney, and third author Jan-Niklas Boyn found that they could add a universal correction to density functional theory that allows the electrons to become entangled among multiple orbitals at once.
"This allows the orbitals in the calculation to be not only fully filled or fully empty, but anywhere in between," said Mazziotti. "We arrive at a one-electron picture that is still able to capture the behavior that arises from correlated many-body electron effects."
A 'universal' adaptation
As a bonus, the scientists said, the code can be added to existing algorithms without having to rewrite that code. "Basically, the correction kicks in whenever it's needed, but doesn't interfere with the rest of the code otherwise," said Gibney.
It's also universal—in that it can be added to code simulating many kinds of electronic behavior, be it photovoltaic solar panels or carbon sequestration or superconducting materials—or even biology.
For example, explained Boyn, one application could be in understanding the chemistry that takes place using enzymes containing metal atoms, known as metalloenzymes.
"There is a plethora of metalloenzymes responsible for a lot of the chemistry in your cells, for example, but they have been notoriously hard to describe with current models," he said. "This theory could in the near future allow us to tackle this chemistry in a way that is impossible right now."
More information: Daniel Gibney et al, Universal Generalization of Density Functional Theory for Static Correlation, Physical Review Letters (2023). DOI: 10.1103/PhysRevLett.131.243003
Journal information: Physical Review Letters
Provided by University of Chicago