This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study identifies key ingredient for affordable fuel cell catalysts
To make fuel cells more affordable, researchers have spent decades searching for low-cost catalysts to replace platinum and other expensive metals.
This includes experimenting with different combinations of three abundant and relatively cheap materials: iron, nitrogen and carbon. Results thus far have been uneven. Researchers can make the iron-nitrogen-carbon catalyst durable or efficient, but not both.
A new University at Buffalo-led study may offer a solution. In the journal Nature Catalysis, researchers report how adding hydrogen to the fabricating process creates a strong and effective catalyst that approaches the performance of platinum.
The advancement suggests an important step toward helping fuel cell technology live up to its potential as a pollution-free provider of electricity for cars, trucks, trains, airplanes and other heavy-duty vehicles.
"For years, the scientific community has struggled to balance this tradeoff. We can make low-cost that are effective but degrade too easily. Or we've made them very stable, but their performance couldn't match platinum. With this work, we've taken a step toward solving this problem," says the study's corresponding author, Gang Wu, Ph.D., professor in the Department of Chemical and Biological Engineering in the School of Engineering and Applied Sciences.
The work builds on previous research led by Wu which described iron-nitrogen-carbon catalysts that, while durable, struggled to speed up important chemical reactions within fuel cells.
The new study addressed this limitation during a fabricating process called pyrolysis, which involves using extremely hot temperatures to combine materials.
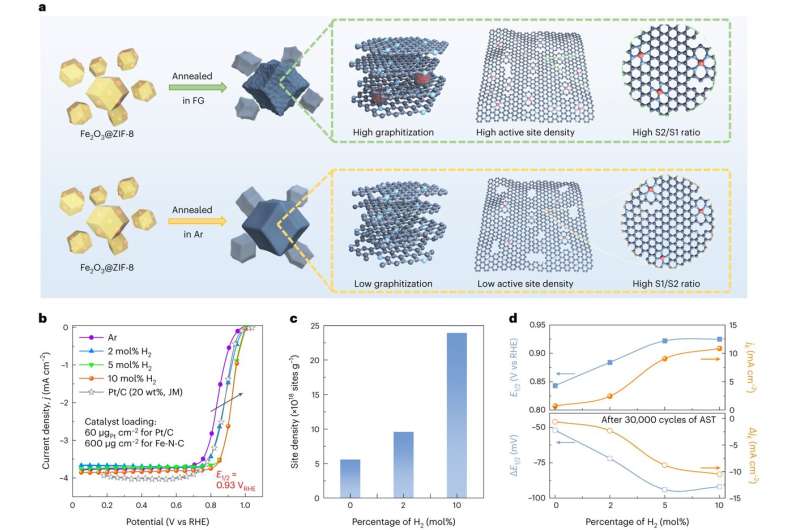
During pyrolysis, researchers bonded four nitrogen atoms to the iron in a high-temperature chamber. They then embedded this material into a few layers of graphene, which is a tough, light and flexible form of carbon.
Usually this process occurs within a chamber featuring an inert gas, such as argon. However, this time researchers fed hydrogen into the chamber to create mixture of 90% argon and 10% hydrogen.
As a result, researchers were able to more precisely control the makeup of the catalyst. Specifically, they were able to place two different iron-nitrogen-carbon compounds (one contained 10 carbon atoms, the other contained 12 carbon atoms) in positions that support durability and efficiency.
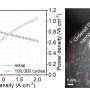
The resulting catalyst reached initial fuel cell performance well beyond the Department of Energy's goal for 2025. It also proved more durable than most iron-nitrogen-carbon catalysts, approaching a typical low-platinum cathode used for fuel cells.
More information: Tuning the thermal activation atmosphere breaks the activity–stability trade-off of Fe–N–C oxygen reduction fuel cell catalysts. Nature Catalysis (2023). DOI: 10.1038/s41929-023-01062-8
Journal information: Nature Catalysis
Provided by University at Buffalo