This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study looks at RNA's solo act on the ever-changing stage of cellular dynamics

RNA has been in the limelight for its starring role in cutting-edge vaccine technology, but RNA molecules are also key players in the inner workings of cells.
This lesser explored RNA exploit is the subject of a new University at Buffalo-led study published Nov. 6 in Nature Chemistry.
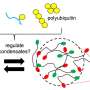
The work examines the role that temperature plays as RNA molecules undergo phase separation to form physically distinct, gel-like condensates. These condensates are specialized, membraneless structures that are involved in various cellular processes and have been associated with neurodegenerative disorders.
Ultimately, the study could help lead to new ways of thinking about biology, biophysics and other fields of study.
"The phase separation of biomolecules has sort of revolutionized our thinking of how cells compartmentalize processes," says Priya Banerjee, Ph.D., associate professor in the UB Department of Physics, within the College of Arts and Sciences, who led the study.
"Most studies have been protein-centric, with the idea being that proteins form these liquid-like condensates, and we have been very interested in what RNA does to this process. So far, studies have been limited to examining how RNA can regulate protein phase separation, so looking at RNA in more of a regulatory role."
The study was done in collaboration with Rohit Pappu, Ph.D., the Gene K. Beare Distinguished Professor of biomedical engineering at Washington University in St. Louis, and Venkat Gopalan, Phd, professor of chemistry and biochemistry at Ohio State University.
Investigating RNA's solo act
Banerjee and Gable Wadsworth, a postdoctoral researcher in Banerjee's lab and first author of the study, became intrigued by how RNA might break out of its regulatory role and phase separate on its own. Through a systematic sleuthing effort, they concluded that all RNA molecules appear to have lower critical solution temperature (LCST) phase behavior, where phase separation is favored at high temperatures. What really surprised them, though, was that polyphosphate, the RNA backbone devoid of the nucleobases and the ribose group, also showed LCST phase behavior.
To get to the bottom of this observed phenomenon, Banerjee and Wadsworth teamed up with Pappu and his group to understand the mechanisms underpinning the behavior.
"We used computations and some of our theoretical understanding of LCST phase behaviors and realized that what Banerjee and colleagues were observing was a combination of two processes," Pappu says. "The phosphate backbone and the solution ions dissolve with increased temperature. The loss of hydration waters from complementary halves drives RNA molecules to seek one another, and the ions bridge phosphate groups within and across different molecules to enable phase separation."
As a result, the condensed phases become physically crosslinked networks, and together, Pappu's and Banerjee's groups worked out that the networking afforded by strong interactions among RNA molecules can enable different phase behavior when heating or cooling. Notably, the team found that dropping the temperature can lead to persistent condensates. Banerjee's lab also worked with Gopalan's lab to understand how condensate formation and the interplay between phase separation and percolation impact the functions of an ancient RNA enzyme.
"RNA has this interesting thermometer, if you will, that senses temperature change," Banerjee says. "This study is a new direction in how we think about phase separation of molecules in general, and could lead to a new understanding of biology, biophysics, materials science and even the origins of life."
Pappu adds that he envisions using RNA's thermoresponsive phase behavior in a range of applications from memory processing and storage to biomaterials.
More information: Gable M. Wadsworth et al, RNAs undergo phase transitions with lower critical solution temperatures, Nature Chemistry (2023). DOI: 10.1038/s41557-023-01353-4
Journal information: Nature Chemistry
Provided by University at Buffalo