How chimeric, droplet-forming proteins may contribute to cancer
In the prelude to cancer, genetic mutations can sometimes lead to the formation of chimeric proteins made from parts of two different proteins that are usually separate.
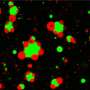
A new review paper examines the latest science on a certain class of these cancer-linked "oncofusion proteins," focusing on those oncofusion proteins that can form liquid-like droplets called "ectopic condensates" in cells.
The article was published on April 25 in Trends in Cell Biology by University at Buffalo researchers Richoo B. Davis, Mahdi Muhammad Moosa, and Priya R. Banerjee, all in the Department of Physics in the UB College of Arts and Sciences. Banerjee, Ph.D., is an assistant professor of physics, and Davis, Ph.D., and Moosa, Ph.D., are postdoctoral associates in the Banerjee Lab.
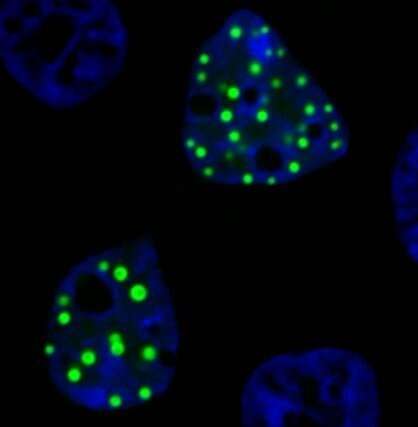
In laboratory experiments, ectopic condensates emerge when part of a protein that normally forms droplets is fused with part of another protein usually found in a different location within a cell. The resulting chimeric protein retains some functions from both of the original proteins, and is capable of forming droplets in the "wrong" place inside cells, Moosa says.
Protein droplets, also known as "membrane-less organelles," can serve as hubs of biochemical activity, so misplaced droplets with aberrant functions are interesting to investigate, the researchers say.
"One important audience for our review is cancer researchers," Moosa says. "Biophysicists who study biomolecular condensates may already be familiar with these concepts, but we wanted to reach out and share these insights with cancer biology researchers who are directly working with patient samples."
Davis notes that ectopic condensates could offer an attractive target for cancer therapeutics, but that more research is needed to better understand how these chimeric proteins operate in their natural state and how they may rewire gene transcription that leads to cancer development: "The cell is a very complex system," he says. "We need better tools to study protein condensates in their native conditions, and our future studies will be focused to address this."
In the review article, the authors summarize emerging findings from multiple recently published works from different research groups, including a 2021 study they completed along with Taranpreet Kaur, a recent UB physics Ph.D. graduate.
That earlier paper, appearing in a special issue of the journal Protein Science on "Biophysics of Biomolecular Condensates," focused on a FET family oncofusion protein and was titled, "FUS oncofusion protein condensates recruit mSWI/SNF chromatin remodeler via heterotypic interactions between prion-like domains." The team showed how these ectopic protein condensates can recruit key molecular machines to change the balance of gene regulation.
"The discovery of protein phase separation has changed our view of how cells organize their internal space," Banerjee says. "As more and more research emerges in this exciting field, we are learning about the role of protein droplets in key biological processes such as gene regulation and their roles in fatal human diseases. Based on the emerging data from different laboratories, we suspect that a subset of cancer-linked fusion proteins can form new types of condensates, which normal cells don't possess."
"The study of naturally occurring fusion proteins and their condensates with new biological functions are not only important from the cancer biology perspective but also from the viewpoint of protein engineering, as learning how these fusion proteins work will provide new opportunities to create artificial proteins with novel applications in biomedical sciences," Banerjee adds.
More information: Richoo B. Davis et al, Ectopic biomolecular phase transitions: fusion proteins in cancer pathologies, Trends in Cell Biology (2022). DOI: 10.1016/j.tcb.2022.03.005
Richoo B. Davis et al, FUS oncofusion protein condensates recruit mSWI/SNF chromatin remodeler via heterotypic interactions between prion‐like domains, Protein Science (2021). DOI: 10.1002/pro.4127
Provided by University at Buffalo