This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers discover how to prevent formaldehyde from inhibiting hydrogen-producing enzymes
Enzymes from microorganisms can produce hydrogen (H2) under certain conditions, which makes them potential biocatalysts for biobased H2 technologies. In order to make this hydrogen production efficient, researchers are trying to identify and eliminate possible limiting factors. These include formaldehyde, which occurs naturally as a metabolic product in cells and inhibits the particularly efficient [FeFe] hydrogenase.
A team from the work group Photobiotechnology at Ruhr University Bochum, Germany, was able to elucidate and switch off this underlying mechanism. The researchers have reported their findings in the Journal of the American Chemical Society on 20 November 2023.
Formaldehyde is known as a preservative, among other things, but it also occurs as a natural metabolite in living cells. Twelve years ago, scientists from the University of Oxford, U.K., and Ruhr University Bochum, Germany, showed that this omnipresent molecule inhibits a certain class of biocatalysts, namely the particularly efficient hydrogen-generating hydrogenases of the two-iron type—so-called [FeFe]-hydrogenases.
"This was an interesting discovery, because formaldehyde could inhibit both the natural H2 metabolism of microorganisms and isolated hydrogenases in biotechnological applications," explains Dr. Jifu Duan, first author of the study.
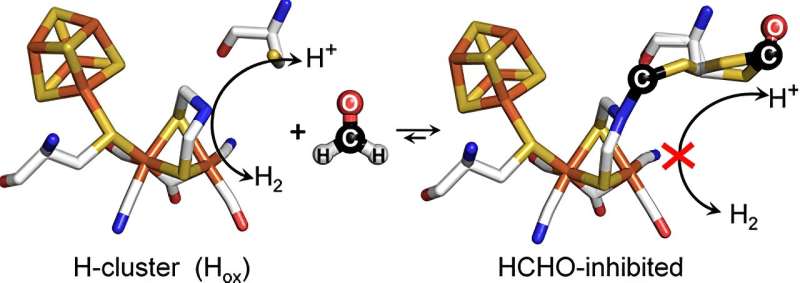
After various theoretical studies had hypothesized how the formaldehyde molecule might influence [FeFe]-hydrogenases, a team of researchers led by Duan and Professor Eckhard Hofmann at Ruhr University has now succeeded in elucidating the molecular mechanism experimentally. Using structures of formaldehyde-treated [FeFe]-hydrogenases obtained by protein crystallography, they were able to show that formaldehyde reacts with the so-called active center of the biocatalysts—an inorganic protein part where protons and electrons are converted to H2.

In addition, however, formaldehyde combines with another very important protein part, which is necessary for the transport of protons to the active center by means of a sulfur-containing chemical group. When the scientists replaced this part with another, formaldehyde was hardly able to exert its inhibitory effect.
"Future biotechnological applications of [FeFe]-hydrogenases may well involve the presence of formaldehyde, so that our modified formaldehyde-resistant biocatalysts could be used here," explains Duan. "We also believe that our findings can be transferred to other biocatalysts." This could play a role for bio-based industrial processes, but also for understanding metabolic pathways in living organisms.
More information: Jifu Duan et al, Insights into the Molecular Mechanism of Formaldehyde Inhibition of [FeFe]-Hydrogenases, Journal of the American Chemical Society (2023). DOI: 10.1021/jacs.3c07800
Journal information: Journal of the American Chemical Society
Provided by Ruhr-Universitaet-Bochum