This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Majority rule in complex mixtures: Study identifies new mechanism for control of phase separation
The very first life on Earth is thought to have developed from "protocells"—liquid mixtures of many different types of molecules. Researchers from the University of Göttingen have now shown that in such mixtures, small imbalances in the number of molecules of different types can have an unexpected effect.
A surprising interplay with the complex pattern of interactions strongly amplifies such imbalances—meaning that a type of molecule that is only slightly in the majority can almost entirely separate out from the others. These fundamental findings point towards a new mechanism that will apply in many complex mixtures. For instance, this would be a way for cells to control the formation of structures by fine-tuning the concentration of different molecules. The research was published in Physical Review Letters.
When living cells first appeared, structures formed via liquids separating into distinct regions, in a process known as "phase separation." In the same way that a vinaigrette (a mixture of vinegar and oil) separates into droplets if left to sit after stirring, phase separation created structures in the cell that evolved to become what are known in the cells alive today as "membraneless organelles."
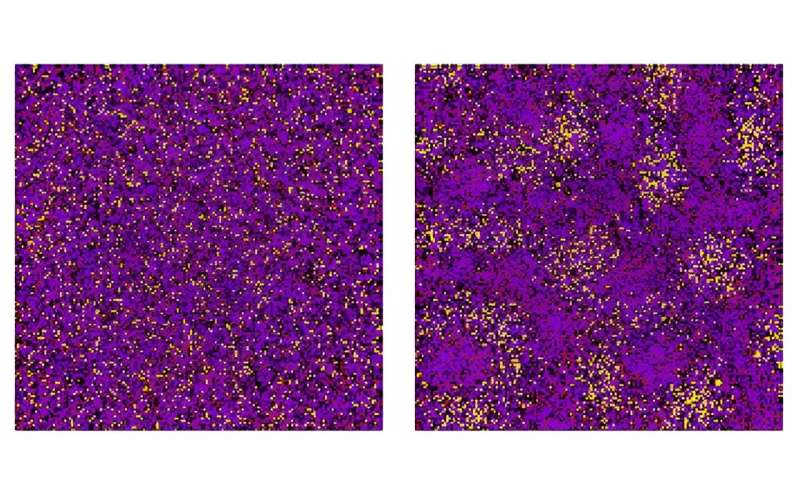
The researchers used a mathematical model for the complex interactions between the many different types of molecules in a mixture. Their model incorporates information on the composition, meaning the amount of each component of the mixture. They applied the laws of thermodynamics to identify if the liquid mixture will undergo phase separation and what kind of structures will be formed.
In the past, due to the complexity of the problem, only uniform compositions could be studied, assuming an equal number of molecules of each type. The researchers from the University of Göttingen developed a more sophisticated method to take this into account and discovered that small imbalances in the composition—that is, small deviations from a uniform composition—can make a big difference via thermodynamic instabilities. This can lead to a small number of mixture components driving the formation of new phases.
"It's surprising that even very small imbalances in the composition can be strongly amplified. For simple interactions, entropy would be maximized by creating new phases of similar composition. But here, even if there is just 2% of one component in a mixture of 100 different molecules, that component can be present at a concentration of 70% in a droplet that is beginning to separate," comments lead author Filipe Thewes, Ph.D. researcher at Göttingen University's Institute for Theoretical Physics.
This amplification could provide a mechanism for cells to regulate phase separation and thus structure formation, simply by making small changes in their composition.
Senior author Professor Peter Sollich, also at Göttingen University's Institute for Theoretical Physics, adds, "Because our mathematical approach can be applied generally to many different situations, these results offer exciting potential for applications in related models. For instance, this mechanism could be at work in a range of different fields—from living cells to market economies or ecological networks with many species."
More information: Filipe C. Thewes et al, Composition Dependent Instabilities in Mixtures with Many Components, Physical Review Letters (2023). DOI: 10.1103/PhysRevLett.131.058401
Journal information: Physical Review Letters
Provided by University of Göttingen