This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Exploring the self-organizing origins of life
Catalytic molecules can form metabolically active clusters by creating and following concentration gradients—this is the result of a new study by scientists from the Max Planck Institute for Dynamics and Self-Organization (MPI-DS). Their model predicts the self-organization of molecules involved in metabolic pathways, adding a possible new mechanism to the theory of the origin of life.
The results can help to better understand how molecules participating in complex biological networks can form dynamic functional structures, and provide a platform for experiments on the origins of life.
One possible scenario for the origin of life is the spontaneous organization of interacting molecules into cell-like droplets. These molecular species would form the first self-replicating metabolic cycles, which are ubiquitous in biology and common throughout all organisms. According to this paradigm, the first biomolecules would need to cluster together through slow and overall inefficient processes.
Such slow cluster formation seems incompatible with how quickly life has appeared. Scientists from the department of Living Matter Physics from MPI-DS have now proposed an alternative model that explains such cluster formation and thus the fast onset of the chemical reactions required to form life.
"For this, we considered different molecules, in a simple metabolic cycle, where each species produces a chemical used by the next one," says Vincent Ouazan-Reboul, the first author of the study. "The only elements in the model are the catalytic activity of the molecules, their ability to follow concentration gradients of the chemicals they produce and consume, as well as the information on the order of molecules in the cycle," he continues.
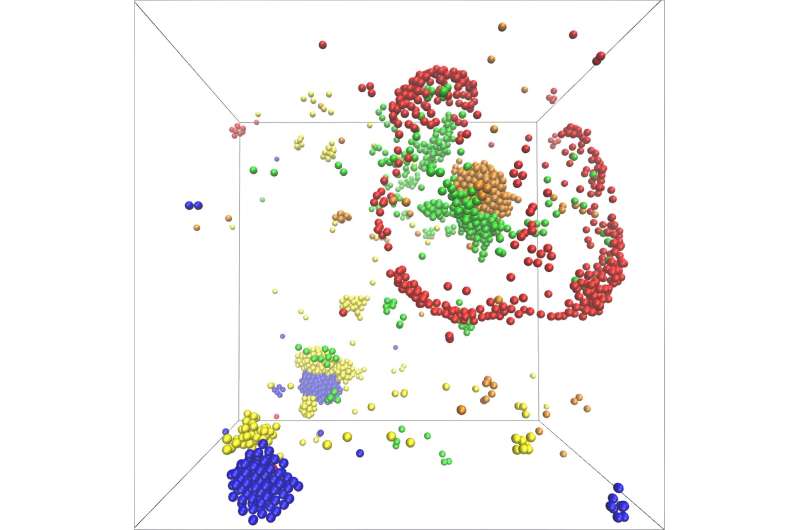
Consequently, the model showed the formation of catalytic clusters including various molecular species. Furthermore, the growth of clusters happens exponentially fast. Molecules hence can assemble very quickly and in large numbers into dynamic structures.
"In addition, the number of molecule species which participate in the metabolic cycle plays a key role in the structure of the formed clusters," Ramin Golestanian, director at MPI-DS, summarizes, "Our model leads to a plethora of complex scenarios for self-organization and makes specific predictions about functional advantages that arise for odd or even number of participating species. It is remarkable that non-reciprocal interactions as required for our newly proposed scenario are generically present in all metabolic cycles."
In another study, the authors found that self-attraction is not required for clustering in a small metabolic network. Instead, network effects can cause even self-repelling catalysts to aggregate. With this, the researchers demonstrate new conditions in which complex interactions can create self-organized structures.
Overall, the new insights of both studies add another mechanism to the theory of how complex life once emerged from simple molecules, and more generally uncover how catalysts involved in metabolic networks can form structures.
The paper is published in the journal Nature Communications.
More information: Vincent Ouazan-Reboul et al, Self-organization of primitive metabolic cycles due to non-reciprocal interactions, Nature Communications (2023). DOI: 10.1038/s41467-023-40241-w
Journal information: Nature Communications
Provided by Max Planck Society