This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
One-pot synthesis of iron–sulfur proteins under aerobic conditions

S clusters, which are a part of Fe–S proteins, are found across all forms of life. They play a significant role as biological cofactors—helper molecules that assist these proteins in different biochemical transformations—that are involved in respiration and metabolism.
These clusters are of keen research interest since they are considered to be a critical part of evolution. They serve as a link between pre-biotic chemistry (chemical processes that existed prior to the emergence of life forms) and the complex molecular and biological systems we know today. Put simply, they might be one of the primitive catalysts that led to the emergence of life on Earth. Thus, having convenient methods to synthesize Fe–S proteins will hopefully advance our understanding of young Earth biology and help us answer the ultimate question of the origin of life.
However, despite their prevalence, the synthesis of mature Fe–S proteins outside the cell has proven to be challenging. They not only require intricate cellular machinery for synthesis but also degrade easily upon contact with oxygen due to its reaction with their Fe–S clusters.
Therefore, scientists had been forced to follow the complicated route of first producing and extracting an incomplete (or 'apo') protein, followed by its maturation (addition of the Fe–S cofactor) under strictly oxygen-deprived conditions. But what makes this process even harder is the presence of contaminating iron-containing proteins in the final extract.
A team of researchers, including Associate Professors Kosuke Fujishima and Shawn McGlynn from the Earth-Life Science Institute (ELSI), Tokyo Institute of Technology and Assistant Professor Po-Hsiang Wang from the National Central University have developed a novel protocol for producing mature [4Fe-4S] proteins in which the Fe–S cluster is placed within a cube-like structure.
The team devised a specialized Fe–S assembly protein system pathway that would function in an oxygen-free environment due to the presence of an oxygen-scavenging system to deliver mature Fe–S proteins. The paper is published in the journal ACS Synthetic Biology.
The researchers first sought to assemble what's known as the sulfur formation (SUF) system. In bacteria, this multi-protein system contains all the necessary machinery to produce [4Fe-4S] clusters. It has a higher tolerance for oxygen when compared to the other pathways with similar functions (such as nitrogen fixation and the iron–sulfur cluster system). The research team created a recombinant SUF pathway that consists of six protein subunits with the ability to function in a cell-free environment.
In order to maintain an oxygen-free environment within the test tube, the researchers then introduced a three-enzyme cascade (a set of three enzymatic reactions that occur in a sequence) that serves as an oxygen-scavenging system. While this scavenging system removes oxygen from the environment, it also improves the efficiency of the system. It achieves this by producing reduced flavin adenine dinucleotide (FADH2), an electron-carrier which is needed for the synthesis of Fe-S cluster by SUF system.
Finally, for the synthesis of apo protein, the team adopted a specialized cell-free method that enables the in vitro production of proteins by using reconstituted cell-free protein synthesis known as PURE system. With the addition of genetic material (DNA or mRNA) and the necessary sources of energy, the PURE system essentially acts as an artificial protein factory.
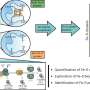
![This diagram shows the two main steps involved in the proposed synthesis protocol. Step 1 shows the chemical cascade used to create an oxygen-free environment, alongside the PURE system used to synthesise the 'immature' (apo) proteins. Step 2 shows the implementation of the SUF machinery, which adds the [4Fe–4S] cluster to the apo proteins, yielding functional and mature [4Fe–4S] proteins. Credit: Reproduced from Wang and Nishikawa et al. 2023 ACS Synthetic Biology Overcoming the challenges to synthesising iron–sulfur proteins outside the glovebox](https://scx1.b-cdn.net/csz/news/800a/2023/overcoming-the-challen.jpg)
Thus, the researchers combined the PURE system, the O2-scavenging enzyme cascade, and the components of the SUF pathway in a single tube to perform one-pot, cell-free synthesis of two representative [4Fe-4S] proteins: aconitase and thermophilic ferredoxin.
Explaining the iterative processes experienced in getting the pieces of the puzzle to fit together, a co-first author Ph.D. student Shota Nishikawa comments, "It was challenging to home-on to the appropriate stoichiometry and substrate/cofactor concentration. Nevertheless, we performed thorough studies to characterize the proper ratio of enzymes needed in the PURE system."
The protocol developed in this study has significant implications for the scientific community. "We have created a novel and convenient method for the synthesis of mature Fe–S proteins which no longer requires a bulky glove box," says Fujishima. He claims that their protocol overcomes the traditional challenges of the [4Fe–4S] cluster assembly and O2 sensitivity, which have been major obstacles in the fields of synthetic biology and anaerobic enzymology.
By greatly extending the capabilities of the PURE system, the strategy proposed by the researchers could lead to the development of new biotechnologies and a better understanding of the fundamentals of protein synthesis and assembly.
With eyes on the future, the researchers note that this work opens several doors for upcoming studies. Being able to implement in vitro oxygen-free environments could help scientists replicate other types of multi-protein pathways, such as the nitrogen fixation (NIF) andiron–sulfur cluster (ISC) pathways, to synthesize other metal cofactor-bearing enzymes. This, in turn, may lead to the development of new biocatalysts and synthetic cells, with potential applications in environmental remediation, energy production, medicine, and astrobiology.
More information: Po-Hsiang Wang et al, One-Pot De Novo Synthesis of [4Fe-4S] Proteins Using a Recombinant SUF System under Aerobic Conditions, ACS Synthetic Biology (2023). DOI: 10.1021/acssynbio.3c00155
Journal information: ACS Synthetic Biology
Provided by Tokyo Institute of Technology