This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Researchers identify motor complex of cell division as a promising target for anti-cancer drug design

A biologist at the University of Hawai'i at Hilo, who is doing research into the mechanisms of cell division to find clues for cancer treatment, has just published his most recent findings in the journal Science Signaling.
Associate Professor of Biology Li Tao is a biochemist and cell biologist with expertise in using a combination of in vitro biochemistry and in vivo cell biology to understand the regulation of cell division, thus providing insights into the fundamental mechanism to control the growth of cancer cells.
"Cell division is of tremendous interest to the biomedical research community due to its connection to various human diseases, including cancer," explains Tao. "Understanding the regulation of cell division is crucial to combating cancer. Cytokinesis, as the last stage of cell division, represents the final opportunity to control this process."
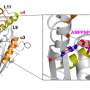
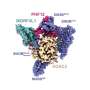
The recently published findings from Tao's lab highlight that centralspindlin, a motor complex implicated in cell division, plays a pivotal role in dictating cytokinesis by transporting essential components required to initiate this process.
"This discovery positions centralspindlin as an ideal target for anti-cancer drug designs," says Tao.
The study was conducted primarily by Tao and Brandt Warecki, who holds a joint postdoctoral position between UH Hilo and University of California, Santa Cruz. Warecki is lead author of the study; Tao serves as corresponding author.
Finding clues for cancer treatment
Tao says any mistakes that escape from cytokinesis will cause severe human diseases including cancer. Several years ago, he discovered that a key motor protein for cytokinesis, kinesin-6, is regulated by Rho-family protein RacGAP. The findings were published in the journal Nature Communications with Tao as both lead author and corresponding author.
In that study, Tao challenged a widely accepted concept in the field: that all kinesins can move along microtubules by themselves. "However," he says, "we found that kinesin-6 alone is not active, meaning it cannot move on microtubules. Kinesin-6 has to bind to another protein RacGAP to activate its function. This finding provides a novel mechanism on the regulation of cytokinesis." After this finding, Tao and his research team continued their study of kinesin-6 and other motor proteins.
Tao says his research into the mechanisms of cell division will provide clues for cancer treatment.
"Abnormal cell division causes cancer," he explains. "Understanding the mechanism of cell division and its control has thus become a key to find cures for cancer." This is why his studies are directly related to the regulation of cell division. His ultimate goal is to have a greater understanding of how mitotic motors regulate cell division, which will help find an effective way to cure cancer.
Tao believes the success of this mission depends on collaboration among peers. "In my opinion, solving problems on cell division should be teamwork with experts from various fields," he says.
More information: Brandt Warecki et al, Centralspindlin-mediated transport of RhoGEF positions the cleavage plane for cytokinesis, Science Signaling (2023). DOI: 10.1126/scisignal.adh0601
Journal information: Nature Communications , Science Signaling
Provided by University of Hawaii at Hilo