This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Towards synthesis of phenanthridine-based pharmaceutical compounds
Phenanthridines are heterocyclic compounds consisting of two six-membered benzene rings fused to a six-membered ring containing nitrogen. They are found in many naturally occurring organic compounds known for their anticancer and antitumor properties. Due to their potential medicinal applications, there is a significant interest in synthesizing phenanthridine derivatives in laboratories.
A promising synthesis approach involves radical isonitrile insertion to produce imidoyl radical intermediates, which then cyclize to form phenanthridine. However, the exact mechanism of isonitrile insertion is not well understood.
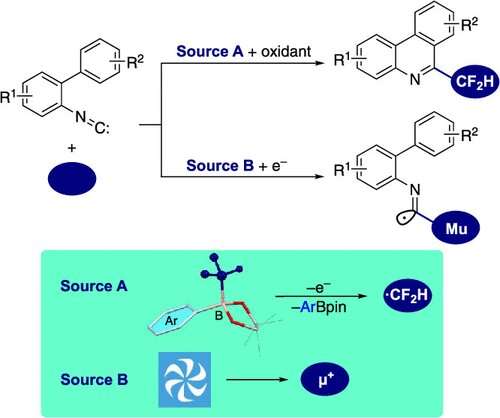
Recently, a team of researchers, led by Associate Professor Shigekazu Ito from Tokyo Institute of Technology (Tokyo Tech), has investigated the use of aryl-substituted difluoromethylborates for synthesizing difluoromethylated phenanthridines. Their study, published in The Journal of Organic Chemistry, assesses the scope of producing pharmaceutically relevant fluorinated phenanthridines from aryl-substituted difluoromethylborates and elucidates the reaction mechanism underlying isonitrile radical addition.
"Taking into account the significance of difluoromethylated phenanthridines in drug discovery, it is desirable to develop novel and complementary synthetic methods for producing 6-(difluoromethyl)phenanthridines, especially via radical isonitrile insertion," points out Dr. Ito.
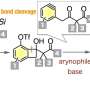
The researchers synthesized 6-(difluoromethyl)phenanthridines by first generating a highly reactive difluoromethyl radical (•CF2H) through the oxidation of aryl-substituted difluoromethylborates. This radical served as the starting point for the isonitrile insertion and cyclization processes within the isonitrile group.
After screening various oxidizing conditions, the researchers identified a combination of silver oxide (Ag2O) and potassium peroxodisulfate (K2S2O8) as ideal initiators for radical isonitrile insertion in 2-isocyano-1,1'-biphenyls. They observed that K2S2O8 oxidizes Ag2O, which, in turn, oxidizes the aryl-substituted difluoromethylborates, leading to the generation of the •CF2H radical. It attaches to the isonitrile group, producing the imidoyl radical, which then undergoes intramolecular cyclization, ultimately leading to the formation of 6-(difluoromethyl)phenanthridine.
The researchers explored various aryl groups in aryl-substituted difluoromethylborates to maximize the yield of 6-(difluoromethyl)phenanthridine. Among the tested aryl groups, p-diethylamino-phenyl-substituted borate was stable and produced the corresponding phenanthridine with a reasonable yield of 53%.
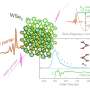
Furthermore, the researchers employed a technique called "transverse-field muon spin rotation" to confirm the reaction mechanism and the presence of the short-lived imidoyl radical. They directed a beam of positive muons (subatomic particles similar to protons but nine times lighter) towards the isonitrile group and carefully observed the changes in their spins.
Muons accompanying electrons, called muoniums, preferentially added to the carbon atom of the isonitrile unit, forming an intermediate that subsequently underwent a cyclization process. This observation provided compelling evidence for the existence of the elusive imidoyl radical.
In the future, the team hopes to explore different approaches to generate the difluoromethyl radical for facilitating the production of difluoromethylated phenanthridines. "Besides chemical oxidation, it might be possible to use photocatalytic and electrochemical methods to synthetically generate the difluoromethyl radical from difluoromethylborates," says Dr. Ito.
In conclusion, this study presents a highly promising pathway for synthesizing 6-(difluoromethyl)phenanthridines, a breakthrough that holds tremendous potential for drug development.
More information: Kakeru Konagaya et al, Difluoromethylborates and Muonium for the Study of Isonitrile Insertion Affording Phenanthridines via Imidoyl Radicals, The Journal of Organic Chemistry (2023). DOI: 10.1021/acs.joc.3c00056
Provided by Tokyo Institute of Technology