This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Revealing how an embryo's cells sync up
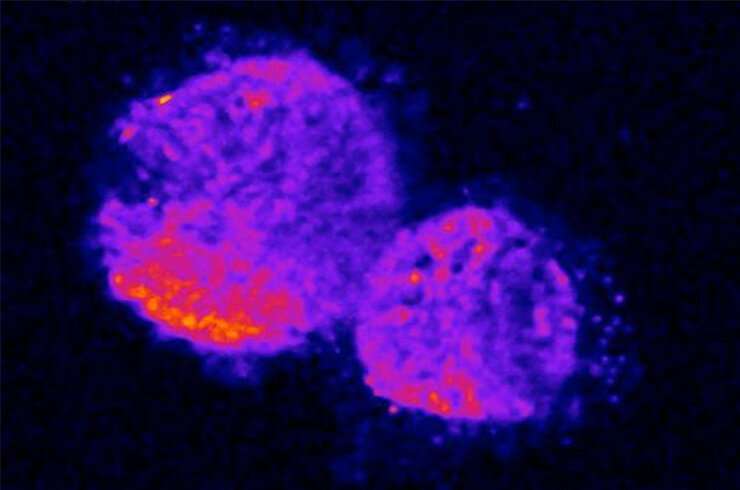
Scientists have known that when a mouse embryo is developing, the cells that will become its spine and muscles switch specific genes on and off repeatedly, in a synchronous fashion. However, there are deep mysteries about how these cells synchronize. FMI researchers have now developed a mathematical model that not only better explains how spontaneous synchronization arises in a developing mouse embryo, but may also offer some fundamental clues about how other biological systems sync up.
From fireflies flashing together to an audience's applause falling into a beat to neurons firing in sync, life offers many examples of spontaneous synchronization across populations. In the developing mouse embryo, for example, synchronous waves of gene-expression patterns across cells drive the formation of tissue segments that then develop into vertebrae and skeletal muscle. While the current go-to model of synchronous systems has captured the basic mechanism underlying this process, it couldn't explain what information cells exchange and what the cells do with the information they get.
Scientists have proposed several possibilities: one cell may slow down its wave of gene expression and another cell would speed it up, so eventually their gene-expression cycles would fall in sync. But researchers led by Charisios Tsiairis found that a different mechanism comes into play. The team discovered that one cell sends a molecular signal that "tells" another cell to pause its gene-expression cycle until the signal-sending cell gets to the same stage of the cycle. Once the signal-sending cell catches up, the two cells will start activating genes in sync.
The molecular signal that allows cells to communicate is Notch, a conserved protein that spans the cell membrane and regulates many processes, including cell proliferation and death. Notch transmits signals from the outside to the inside of a neighboring cell by interacting with another protein called Delta, which is located on the neighboring cell's surface.
"Cells that have Delta only send the information, and those that have Notch only receive it," Tsiairis says. "It's like two people using a walkie-talkie: when one sends a message, the other can receive the message but can't send any information back at the same time."
The discovery, which is detailed in Developmental Cell, stems from a mathematical model developed by Gregory Roth, a staff scientist at the FMI. Georgios Misailidis, a former Ph.D. student in the Tsiairis lab, then validated the model's predictions in tissues from mouse embryos.
"Our model tells us that if you want to synchronize cells, you have to play with the pulses of Notch signaling," Tsiairis says.
The findings may not only help scientists to manipulate cell synchronizations in developing embryos, but could also offer some fundamental clues about how other biological systems, such as neural networks, sync up—opening multiple possibilities for research and medicine.
More information: Gregory Roth et al, Unidirectional and phase-gated signaling synchronizes murine presomitic mesoderm cells, Developmental Cell (2023). DOI: 10.1016/j.devcel.2023.04.002
Journal information: Developmental Cell