This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New technique probes chemistry of enzyme implicated in Alzheimer's and cancer
If serine racemase is the magician that for decades disguised its trick—inverting an amino acid to assist learning and memory but also levitate risks of Alzheimer's and stroke—chemists just snuck a high-res camera behind the curtain.
Led by the University of Nebraska–Lincoln's David Berkowitz and Stephany Ramos de Dios, the team recently revealed a technique capable of recording the enzyme in action, taking notes on its performance, and screening for inhibitors that could keep it from doing harm.
Like many other amino acids, L-serine can often be found residing in the biological nanomachines, or proteins, that are central to the business of running an organism. Around the turn of the 21st century, researchers realized that its mirror image—D-serine, what chemists consider the "opposite hand" of its standard L-configuration—was also at play in people. That made it the first and, to date, the only well-described amino acid of its mirrored kind in human biology.
"If you interviewed all chemistry Ph.D.s across the United States, and you asked them, 'True or false? Human biology involves only L-amino acids, and no D-amino acids,' most would say 'True,'" said Berkowitz, Willa Cather Professor of chemistry at Nebraska. "That's because this is still a fairly new discovery. We're just at the beginning of this field."
The relative obscurity of D-serine belies the emerging evidence of its importance as a signaling mechanism employed by neurons, the cellular messengers of the brain. Recent research suggests that below-average levels of D-serine correspond with schizophrenia. Its overexpression, meanwhile, may contribute to neurodegenerative diseases and cell damage associated with ischemic stroke.
"We don't really have tools to study that," Berkowitz said. "We're trying to give our community those tools. We want a dimmer switch, so you can just dial in the level of (D-serine) activity and study the biology. Small molecules are the best dimmer switches to use to control nature's catalysts.
"There are still a lot of biological questions about the role of D-serine and what happens when you elevate or decrease it."
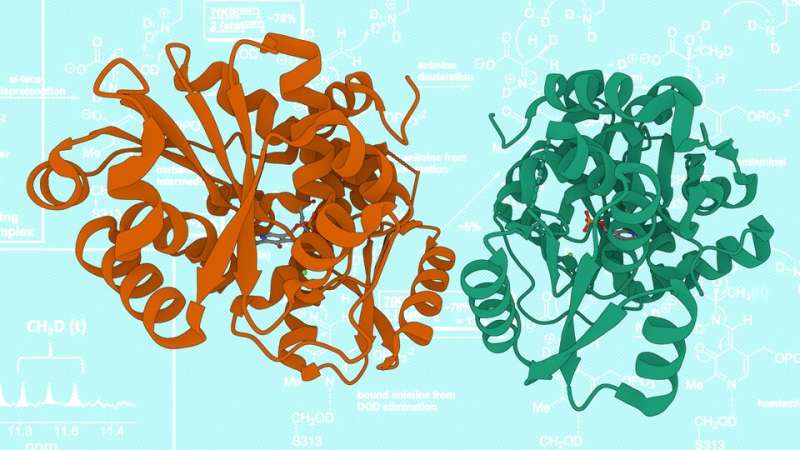
Those early findings and resulting questions have naturally drawn attention to the enzyme, human serine racemase, capable of catalyzing the flip from L-serine to D-. The spotlight grew brighter just last year, when researchers found that the other reaction catalyzed by serine racemase—the conversion of L-serine to a compound called pyruvate—appears to be linked with colorectal cancer.
Now, Berkowitz and six members of his team have developed the first technique that can simultaneously measure both of the enzyme's biologically relevant reactions, allowing users to "watch the chemistry happening in remarkable detail."
To do it, the team relied on the Department of Chemistry's Martha Morton, an expert in nuclear magnetic resonance. Better known as NMR, the technique exposes the nuclei of certain atoms to magnetic fields. By reading the electromagnetic responses of those nuclei, NMR can measure their molecular environment and, by extension, the chemical reactions in which they take part.
So before kickstarting the two chemical reactions with human serine racemase, Morton, Ramos de Dios and doctoral student Jared Hass strategically tagged two atoms in L-serine with carbon-13 isotopes provided by Cambridge Isotope Laboratories. Unlike 99% of the carbon atoms found in nature, carbon-13 isotopes feature a nucleus detectable by NMR.
"This is where organic chemistry meets analytical chemistry," said Berkowitz, who also directs the Division of Chemistry at the National Science Foundation in Washington, D.C. "The organic chemists can surgically place the carbon-13 label in some desired molecule, in a desired position, and when you read the NMR, everything else is pretty much silent, and we're looking only at those two reactions."
Sure enough, the researchers saw the enzyme catalyzing the removal of a hydrogen atom from one side of the serine, followed by its attachment on the opposite side—the mirror-image transition of L- to D-. At the same time, they watched it eject a water molecule and ultimately yield pyruvate. In the process, the team verified that the enzyme actually produces pyruvate substantially faster than it does D-serine.
Even faster, though, was a reaction never before seen by any of the research groups, from the Czech Republic to the United Kingdom, that had begun studying the enzyme—a reaction exchanging the hydrogen in L-serine for deuterium. That normally dormant reaction was "lit up," Berkowitz said, by attaching the carbon-13 labels and performing the chemistry in what's sometimes called heavy water, which contains two deuterium atoms in place of the two lighter hydrogen atoms that typically accompany an oxygen.
"All these pathways start from the same common intermediate (molecule)," Hass said. "So once you establish that intermediate, you can kind of see which avenue the reaction is going to go."
The team was far from finished. Hass joined doctoral alumni Ramos de Dios and Danielle Graham—whose efforts would help land them the department's Cromwell and Hamilton awards, respectively—in assembling a roster of 1,020 chemical compounds. Alongside Nivesh Kumar, a postdoctoral researcher in Berkowitz's lab, the researchers then used the NMR-based technique to test whether any of the compounds might bind to the human serine racemase and, in doing so, inhibit the enzyme's ability to turn L-serine into D-. While the team identified multiple families of inhibitors, one of the 1,020 in particular stood out.
Though that one specific inhibitor—and the family of others Kumar built around it—binds as tightly to the enzyme as any drug-like compound reported so far, the search for candidates will continue, Berkowitz said. Still, early results suggest that the team's most promising candidate affixes itself to a site on serine racemase that would allow it to contend with fewer competitors for the enzyme's real estate, Hass said.
"You should have a finer-tuned modulation of activity," Hass said, "so your dimmer switch is a lot more precise."
More importantly, Berkowitz said, their efforts demonstrated the new platform's ability to facilitate the hunt for, and evaluate the performance of, potential pharmaceutical-grade inhibitors.
"We can (ideally) make inhibitors of this enzyme and then dial them in at different concentrations," he said. "All kinds of biological readouts could be measured as a function of dialing up or dialing down this enzyme. Nobody can really do that yet, including ourselves. But we're working toward that."
The researchers still weren't done. To demonstrate the generality of the approach, Hass and Aina Antony, the newest doctoral student in Berkowitz's lab, tried applying it to another enzyme that represents a key target of the group's ongoing work. Like serine racemase, that enzyme needs a form of vitamin B6 to help it catalyze chemical reactions.
"We did the experiment, and we got great results," Antony said of those efforts, which would help earn the study a place in the Journal of the American Chemical Society.
Of all the chemical reactions in biology that have to be kickstarted by enzymes, roughly 4% of them are catalyzed specifically by vitamin B6-dependent enzymes. To the extent that those enzymes behave in similar ways, the team said, the platform can likely track the reactions spurred by many of them, too.
Berkowitz underscored that all of it—the development of the technique, the demonstration of its ability to track the serine racemase reactions, to screen for inhibitors, to do the same in other enzymes—was possible only through the entire team. And its diversity, whether across three subdisciplines of chemistry or the expanses of the United States, the Caribbean Sea and the Pacific Ocean, should not be overlooked, he said.
"There's another story here," Berkowitz said, "about what kinds of partnerships the university catalyzes. I think Nebraskans are very proud of their university. But I don't think most Nebraskans realize how international (it is) and how broadly represented the country is on our campus. This project is a microcosm of that."
"That's the beauty of the scientific mix," Kumar said. "Everyone doesn't share the same culture or language, but we come and sit and do the same science, with the same motivation."
More information: Stephany M. Ramos de Dios et al, Information-Rich, Dual-Function 13C/2H-Isotopic Crosstalk NMR Assay for Human Serine Racemase (hSR) Provides a PLP-Enzyme "Partitioning Fingerprint" and Reveals Disparate Chemotypes for hSR Inhibition, Journal of the American Chemical Society (2023). DOI: 10.1021/jacs.2c12774
Journal information: Journal of the American Chemical Society
Provided by University of Nebraska-Lincoln