This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Transforming the way cancer vaccines are designed and made
A new way to significantly increase the potency of almost any vaccine has been developed by researchers from the International Institute for Nanotechnology (IIN) at Northwestern University. The scientists used chemistry and nanotechnology to change the structural location of adjuvants and antigens on and within a nanoscale vaccine, greatly increasing vaccine performance. The antigen targets the immune system, and the adjuvant is a stimulator that increases the effectiveness of the antigen.
The study was published Jan. 30 in Nature Biomedical Engineering.
"The work shows that vaccine structure and not just the components is a critical factor in determining vaccine efficacy," said lead investigator Chad A. Mirkin, director of the IIN. "Where and how we position the antigens and adjuvant within a single architecture markedly changes how the immune system recognizes and processes it.
Mirkin also is the George B. Rathmann Professor of Chemistry at the Weinberg College of Arts and Sciences and a professor of medicine at Northwestern University Feinberg School of Medicine.
This new heightened emphasis on structure has the potential to improve the effectiveness of conventional cancer vaccines, which historically have not worked well, Mirkin said.
Mirkin's team has studied the effect of vaccine structure in the context of seven different types of cancer to date, including triple-negative breast cancer, papillomavirus-induced cervical cancer, melanoma, colon cancer and prostate cancer to determine the most effective architecture to treat each disease.
Conventional vaccines take a blender approach
With most conventional vaccines, the antigen and the adjuvant are blended and injected into a patient. There is no control over the vaccine structure, and, consequently, limited control over the trafficking and processing of the vaccine components. Thus, there is no control over how well the vaccine works.
"A challenge with conventional vaccines is that out of that blended mish mosh, an immune cell might pick up 50 antigens and one adjuvant or one antigen and 50 adjuvants," said study author and former Northwestern postdoctoral associate Michelle Teplensky, who is now an assistant professor at Boston University. "But there must be an optimum ratio of each that would maximize the vaccine's effectiveness."
Enter SNAs (spherical nucleic acids), which are the structural platform—invented and developed by Mirkin—used in this new class of modular vaccines. SNAs allow scientists to pinpoint exactly how many antigens and adjuvants are being delivered to cells. SNAs also enable scientists to tailor how these vaccine components are presented, and the rate at which they are processed. Such structural considerations, which greatly impact vaccine effectiveness, are largely ignored in conventional approaches.
Vaccines developed through 'rational vaccinology' offer precise dosing for maximum effectiveness
This approach to systematically control antigen and adjuvant locations within modular vaccine architectures was created by Mirkin, who coined the term rational vaccinology to describe it. It is based on the concept that the structural presentation of vaccine components is as important as the components themselves in driving efficacy.
"Vaccines developed through rational vaccinology deliver the precise dose of antigen and adjuvant to every immune cell, so they are all equally primed to attack cancer cells," said Mirkin, who also is a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. "If your immune cells are soldiers, a traditional vaccine leaves some unarmed; our vaccine arms them all with a powerful weapon with which to kill cancer. Which immune cell 'soldiers' do you want to attack your cancer cells?" Mirkin asked rhetorically.
Building an (even) better vaccine
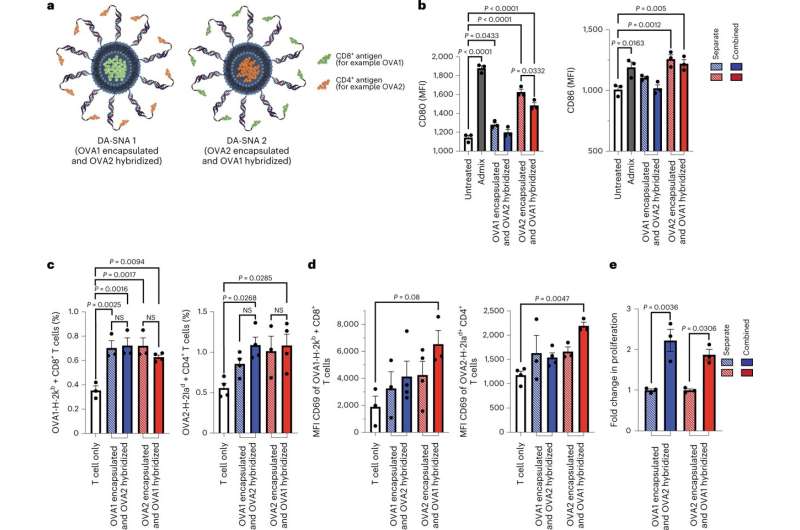
The team developed a cancer vaccine that doubled the number of cancer antigen-specific T cells and increased the activation of these cells by 30% by reconfiguring the architecture of the vaccine to contain multiple targets to help the immune system find tumor cells.
The team investigated differences in how well two antigens were recognized by the immune system depending on their placement—on the core or perimeter—of the SNA structure. For an SNA with optimum placement, they could increase the immune response and how quickly the nanovaccine triggered cytokine (an immune cell protein) production to boost T cells attacking the cancer cells. The scientists also studied how the different placements affected the immune system's ability to remember the invader, and whether the memory was long-term.
"Where and how we position the antigens and adjuvant within a single architecture markedly changes how the immune system recognizes and processes it," Mirkin said.
The most powerful structure throws two punches to outsmart the wily, mutating tumor
The study data show that attaching two different antigens to an SNA comprising a shell of adjuvant was the most potent approach for a cancer vaccine structure. It led to a 30% increase in antigen-specific T-cell activation and doubled the number of proliferating T cells compared to a structure in which the same two antigens were attached to two separate SNAs.
These engineered SNA nanostructures stalled tumor growth in multiple animal models.
"It is remarkable," Mirkin said. "When altering the placement of antigens in two vaccines that are nearly identical from a compositional standpoint, the treatment benefit against tumors is dramatically changed. One vaccine is potent and useful, while the other is much less effective."
Many current cancer vaccines are designed to primarily activate cytotoxic T cells, only one defense against a cancer cell. Because tumor cells are always mutating, they can easily escape this immune cell surveillance, quickly rendering the vaccine ineffective. The odds are higher that the T cell will recognize a mutating cancer cell if it has more ways—multiple antigens—to recognize it.
"You need more than one type of T cell activated, so you can more easily attack a tumor cell," Teplensky said. "The more types of cells the immune system has to go after tumors, the better. Vaccines consisting of multiple antigens targeting multiple immune cell types are necessary to induce enhanced and long-lasting tumor remission."
Another advantage of the rational vaccinology approach, especially when used with a nanostructure like an SNA, is that it's easy to alter the structure of a vaccine to go after a different type of disease. Mirkin said they simply switch out a peptide, a snippet of a cancer protein with a chemical handle that "clips" onto the structure, not unlike adding a new charm to a bracelet.
Path to most effective vaccine for any cancer type
"The collective importance of this work is that it lays the foundation for developing the most effective forms of vaccine for almost any type of cancer," Teplensky said. "It is about redefining how we develop vaccines across the board, including ones for infectious diseases."
In a previously published paper, Mirkin, Teplensky and colleagues demonstrated the importance of vaccine structure for COVID-19 by creating vaccines that exhibited protective immunity in 100% of animals against a lethal viral infection.
"Small changes in antigen placement on a vaccine significantly elevate cell-to-cell communication, cross-talk and cell synergy," Mirkin said. "The developments made in this work provide a path forward to rethinking the design of vaccines for cancer and other diseases as a whole."
Northwestern Ph.D. candidate Michael Evangelopoulos also is an author of the paper, titled "Multi-Antigen Spherical Nucleic Acid Cancer Vaccines."
More information: Chad Mirkin, Multi-antigen spherical nucleic acid cancer vaccines, Nature Biomedical Engineering (2023). DOI: 10.1038/s41551-022-01000-2
Journal information: Nature Biomedical Engineering
Provided by Northwestern University