New methodology for the synthesis of benzazepines
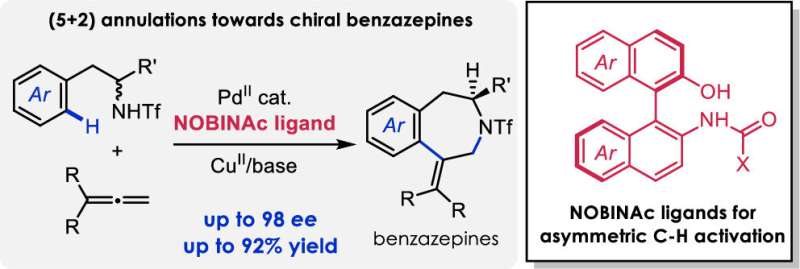
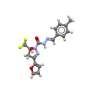
Benzazepines can be found in a wide variety of natural products and pharmaceuticals. Many of these compounds are chiral, being available in two enantiomeric forms. The synthesis of benzazepines with chiral stereocenters represents an ongoing challenge because selective access to these compounds generally requires many steps to successfully achieve.
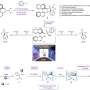
Work recently published in the Journal of the American Chemical Society (JACS) by Center for Research in Biological Chemistry and Molecular Materials (CiQUS) researchers, reports a new methodology that allows the synthesis of these molecules (chiral benzazepines) in a shorter, more efficient and environmentally friendly route.
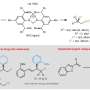
"Our methodology is based on the activation of specific C-H bonds by an organometallic catalyst formed by a palladium metal and a new chiral ligand that has been developed in our laboratory," said Prof. Moisés Gulías, one of the research leaders.
The study, developed by Ph.D. students José Manuel González and Xandro Vidal, and co-directed by Prof. Mascareñas, also includes the collaboration of Dr. Manuel Ortuño, expert in computational chemistry at CiQUS.
The discovery of this new chiral ligand is expected to allow the development of other transformations that involve the enantioselective activation of C-H bonds. In addition, the methodology allows access to libraries of chiral benzazepine derivatives with new biological properties.
More information: José Manuel González et al, Chiral Ligands Based on Binaphthyl Scaffolds for Pd-Catalyzed Enantioselective C–H Activation/Cycloaddition Reactions, Journal of the American Chemical Society (2022). DOI: 10.1021/jacs.2c09479
Journal information: Journal of the American Chemical Society
Provided by Center for Research in Biological Chemistry and Molecular Materials (CiQUS)