A 37% increase to the molecular chiral pool for medicinal chemists expands routes to novel pharmaceuticals
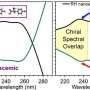
The Cambridge Crystallographic Data Center (CCDC) today highlights that a search of the Cambridge Structural Database (CSD) has found nearly 1,800 conglomerate crystal structures—molecules that have spontaneously enriched chirality upon crystallization—representing a 37% increase on available chiral pool.
These 1,800 structures augment the limited biological chiral pool of synthetic building blocks used by medicinal chemists in drug synthesis, opening new synthetic routes to existing drugs and could lead to new drugs for more effective treatment of disease.
This finding from a team led by Mark Walsh and Matthew Kitching from Durham University, published in JACS Au, introduces a new and potentially unlimited pool of chiral molecules outside of the chiral molecules derived from the natural world—the biological chiral pool. This pool is used by medicinal chemists to introduce chirality to their molecules.
Most biological substances are chiral, including protein binding sites, so drugs that bind to these receptors must also be chiral. Famously the drug Thalidomide was withdrawn from the market when one enantiomer was found to cause birth defects, only 20 years later did scientists find that the other chiral form is safe.
One way used by medicinal chemists to obtain enriched chiral molecules during synthesis is by conglomerate crystallization (where molecules spontaneously crystallize into single enriched crystals) or by using chiral compounds from the limited chiral pool. This research adds a significant alternative source of chirality.
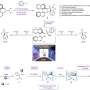
The researchers first mined the ~1.2 million crystal structures in the CSD for those in Sohncke space groups with the potential to be chiral. This produced a subset of over 21,000 crystal candidates that then had their synthesis examined (by reference to the primary literature) to identify those that were produced by conglomerate crystallization. 1800 compounds were identified that were synthesized by racemic methods but spontaneously crystallized in an enriched chiral form.
"We hope that the curation of this list of conglomerate crystals aids the development of preferential crystallization and spontaneous deracemization protocols, while also furthering the understanding of the formation of conglomerate crystal behavior," says Mark Walsh of Durham University.
"I'm delighted to see that the value of the 1.2 million crystal structures in the CSD is once again being utilized in another scientific field beyond crystallography. Medicinal chemists can now use the CSD to greatly widen their options to introduce chirality to their molecules than previously," says Dr. Jürgen Harter, CEO of the CCDC.
More information: Mark P. Walsh et al, Identifying a Hidden Conglomerate Chiral Pool in the CSD, JACS Au (2022). DOI: 10.1021/jacsau.2c00394
Provided by CCDC - Cambridge Crystallographic Data Centre