Researchers access both enantiomers by varying reaction time
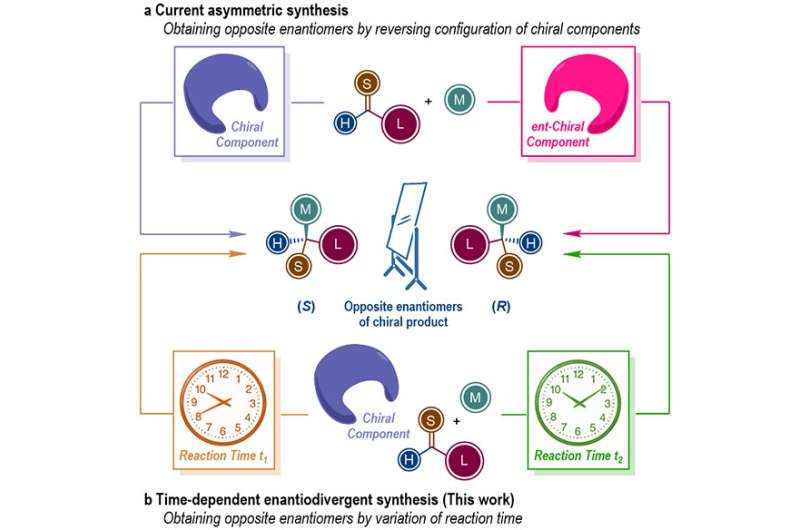
Homochirality is a fundamental feature of all living organism. It is well known that a pair of enantiomers of chiral drug molecules, whose structures have a non-superimposable mirror-image relationship to each other, will probably exert different, or even opposite influences in pharmacological activity, metabolism and toxicity. Therefore, the selective synthesis of a pair of enantiomers of given chiral molecules not only is a prominent task in asymmetric synthesis, but also has significant impacts in pharmaceutical chemistry and materials science.
In general, the preparation of each enantiomer of chiral target molecules requires the reversal of the absolute configuration of certain chiral component in the reaction system. However, many important chiral source molecules only exist in one configuration in nature. Therefore, the preparation of both enantiomers of chiral target molecules via asymmetric synthesis enabled by such chiral sources are quite challenging.
The research group led by Prof. You Shuli at the State Key Laboratory of Organometallic Chemistry, Shanghai Institute of Organic Chemistry recently disclosed a unique phenomenon in asymmetric catalysis where the reaction time is found as the controlling factor to determine the absolute configuration of the chiral products.
More specifically, in the Ir-catalyzed asymmetric allylic amination reactions, the same chiral Ir-complex enables four independent reaction pathways corresponding to the generation and consumption of the (R) and (S) enantiomers of the target allylic amination products.
Thus, the appropriate permutation of the four individual reaction rates guarantees the isolation of each enantiomer of the allylic amination products in a highly enantio-enriched form at different reaction time.
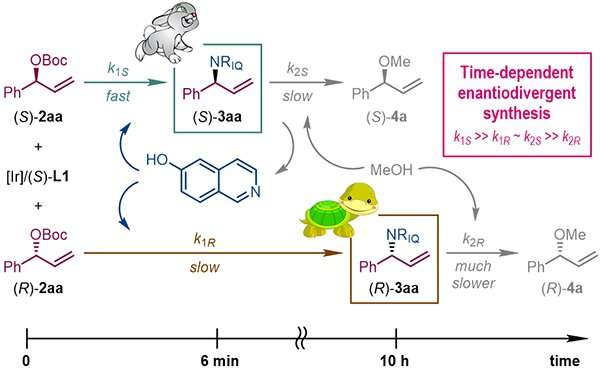
This finding is unprecedented in asymmetric synthesis: it alerts the organic chemistry community to the critical role of reaction time when performing catalytic asymmetric reactions.
The results of this study were published on Nature Chemistry with the title of "Time-dependent enantiodivergent synthesis via sequential kinetic resolution."
More information: Hang-Fei Tu et al. Time-dependent enantiodivergent synthesis via sequential kinetic resolution, Nature Chemistry (2020). DOI: 10.1038/s41557-020-0489-1
Journal information: Nature Chemistry
Provided by Chinese Academy of Sciences