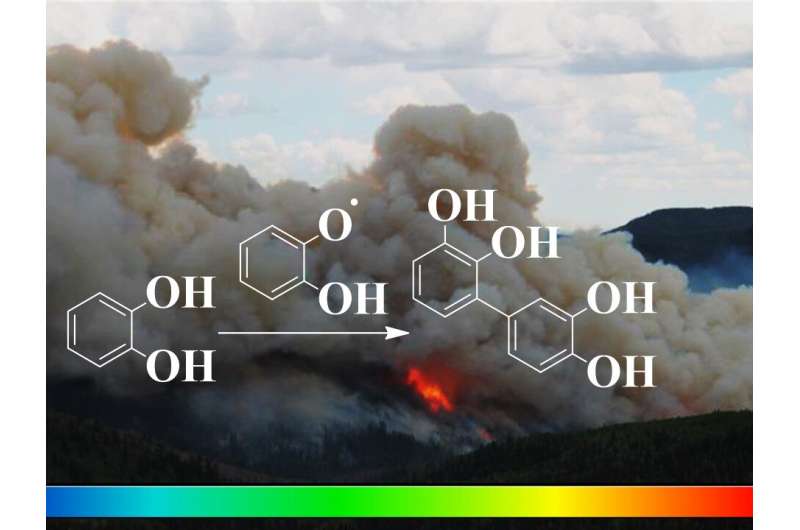
Brown carbon from aromatic pollutants is emitted during combustion and wildfires
Tiny aerosol particles that are suspended in the air can absorb and scatter sunlight radiation and contribute to create clouds affecting climate, reduce the visibility over cities and affect air traffic, and lower air quality. Aerosols in large pollution plumes, called brown clouds, can be transported long distances by the wind and reach other continents from the originating one. The variable composition of particles in brown clouds includes an unhealthy mix of organic molecules and ozone found in smoke.
A laboratory study entitled "Interfacial Oxidative Oligomerization of Catechol" by Dr. Marcelo Guzman and his group at the University of Kentucky now reveals how ozone can transform the organic molecules during surface reactions taking place in such smoke plumes.
The work, published in ACS Omega, reports the detailed coupling reactions of molecules emitted and oxidatively transformed during wildfires, combustion in power plants, and other natural and industrial processes. The researchers determined that aromatic molecules contribute to the formation of light-absorbing oligomers under variable relative humidity, which are important secondary organic aerosols. The significant generation of new dimers and trimers of catechol in these simulated pollution plumes includes the participation of semiquinone radicals in the reaction mechanisms.
When phenols are exposed to background ozone gas and hydroxyl radicals during atmospheric transport, you can expect the formation of some light absorbing oligomers. "We are trying to understand the major transformations of phenols from smoke in the atmosphere, determine their lifetime, and establish how reactions at interfaces contribute to change the chemical composition of pollutants," said Prof. Guzman.
"We would like to develop new understanding of their impacts on air quality and climate. Are the aged molecules more toxic? How do the structural changes of the molecules contribute to create particles that interact with sunlight affecting climate?" A person breathing in these reactive compounds can suffer oxidative damage of cells, especially in the respiratory track and lungs. In addition, these reactive compounds can make some people more prone to other health problems.
Dr. Guzman also states that characterizing the chemical processing of such pollution plumes can help to determine if these brown carbon aerosol derived from wildfires contributes to absorb more heat from the sun or not. "While many small molecules can be photobleached, the larger molecules in the plume may be more resistant to degradation, possibly contributing to warm up the atmosphere," he said.
More information: Marcelo I. Guzman et al, Interfacial Oxidative Oligomerization of Catechol, ACS Omega (2022). DOI: 10.1021/acsomega.2c05290
Journal information: ACS Omega
Provided by University of Kentucky