Organic 2-oxocarboxylic acids provide link to formation of radicals and reactive oxygen species in atmospheric particles

When the humidity in the atmosphere is relatively high, the particles naturally present, also known as aerosol droplets, grow to play a significant role in the chemistry and climate of Earth. These particles are produced from either clean or polluted air, after emissions of gases that nucleate and condense while in the atmosphere. Many times this process is affected by the presence of organic (2-oxocarboxylic) acids that have been characterized to be present over polluted cities.
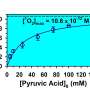
A recent article provides new progress in this field by starting to explain from basic processes such as the dissolution of gas phase molecules into aqueous particles, up to the detailed photochemical pathways that organic molecules undergo in the presence of sunlight. New photochemistry work explains the self-reactions of pyruvic acid and glyoxylic acid, as well as the cross-reactions of pyruvic acid with either glyoxylic acid or glyoxal.
"This is an important contribution to better understand the role that organic molecules play in atmospheric chemistry processes," stated Marcelo Guzman, the principal investigator of the Environmental Chemistry Laboratory at the University of Kentucky. The largest uncertainty in the radiation balance of Earth is related to the roles played by organic aerosols."
Indeed, aerosols also participate in the formation of cloud droplets, and this research interconnects how both cloud formation and aerosol formation are triggered by the aqueous photochemistry of widely spread small 2-oxocarboxylic acids.
This work analyzes the most current scientific findings and opens up the possibility of future atmospheric chemistry research that is needed to improve climate models.
The study is published in the journal Molecules.
More information: Marcelo I. Guzman et al, Aqueous Photochemistry of 2-Oxocarboxylic Acids: Evidence, Mechanisms, and Atmospheric Impact, Molecules (2021). DOI: 10.3390/molecules26175278
Provided by University of Kentucky