August 25, 2022 feature
Bioinspired molecular dyes for biomedical fluorescent imaging
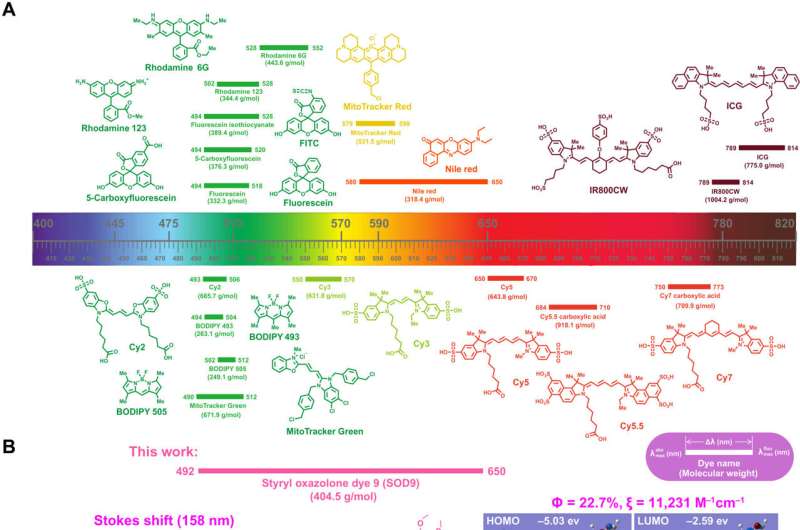
Fluorescence imaging can be conducted with long Stokes shift dyes that minimize crosstalk between the excitation source and fluorescent emission to improve the signal-to-background ratio. Regardless, researchers still form simple, small molecule dyes with large Stokes shift and near infrared emissions. In a new report now published in Science Advances, Hao Chen and a team of scientists developed a series of styrene oxazolone dyes (SODs) using simple synthetic methods inspired by the chromophore chemical structure of fluorescent proteins.
The dyes showed near-infrared emissions with long Stokes shift and small molecular weight. The most promising dyes also showed rapid renal excretion and blood-brain barrier passing properties. The bioengineers modified the compounds for tumor imaging, fluorescence image-guided surgery, neurosurgery and pathological analysis. The findings contribute an essential small molecular dye category to the classical dyes.
Developing long Stokes shift dyes
Fluorescence imaging is widespread in preclinical biomedical research, as well as clinical pathology and fluorescence image-guided surgery. The low cost, easy platform offers minor light damage to the biological specimen for high detection sensitivity. The biomedical application of fluorescent imaging depends on the dyes with critical features, including absorption/emission profiles, absorption coefficient, quantum yield, Stokes shift, and photochemical stability.
Nevertheless, only a few dyes have shown optimal properties across all categories. The heavy crosstalk between the excitation and emission light can result in relatively low signal-to-background ratio. As a result, biochemists aim to develop long Stokes shift near infrared dyes for high signal-to-background ratio. In this work, Chen and the team described the first high-efficiency styrene oxazolone dyes (SODs) as long stokes shift dyes, to provide a new strategy for in vivo fluorescence imaging.
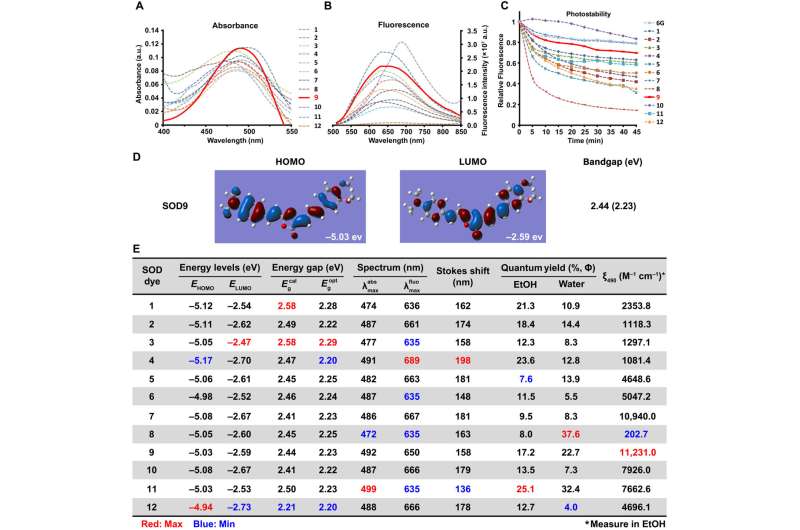
Experiments: Design, synthesis and characterization of styrene oxazolone dyes (SODs)
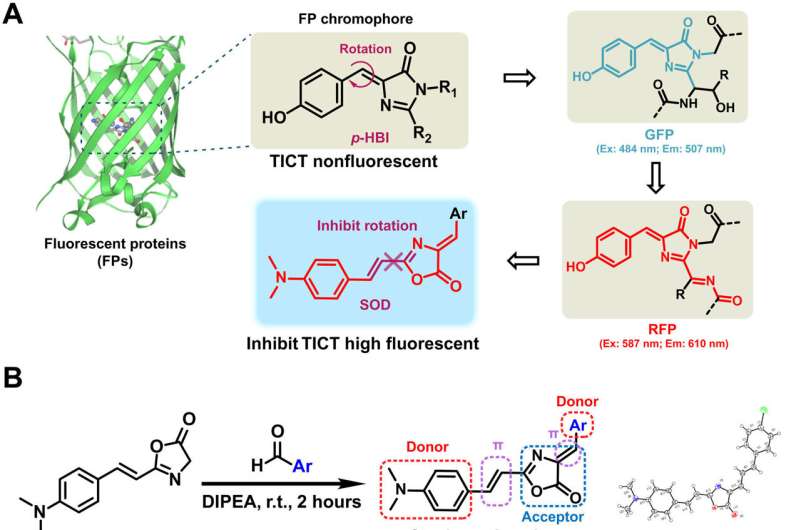
Fluorescence proteins are widely studied in biological research, where they share the same core skeleton of an imidazolinone moiety and an exocyclic double bond to switch between the dark and bright states. The researchers designed and synthesized a series of previously unreported dyes with styrene oxazolone as a fundamental skeleton via a simple procedure at room temperature for good yields within two hours. They characterized the chemical structure of SODs using standard 1H nuclear magnetic resonance (NMR) and high-resolution mass spectrometry spectra. The team identified the spectroscopic properties of the dyes in aqueous media, where they noted strong fluorescence with good photostability, due to the presence of an exocyclic double bond. The researchers derived the difference in spectroscopic properties from the electrical property and position of the substituents.
They thus showed how a variety of factors affected the optical properties of the SODs (styrene oxazolone dyes) and examined the cytotoxicity of the dyes thereafter. They followed these experiments with in vivo pharmacokinetics via fluorescence imaging, as well as in vivo applications of the dye for biomolecule labeling. The results showed how some probes targeting pathological conditions accumulated more in tumor cells than in normal cells, to highlight their tumor-specific targeting properties.
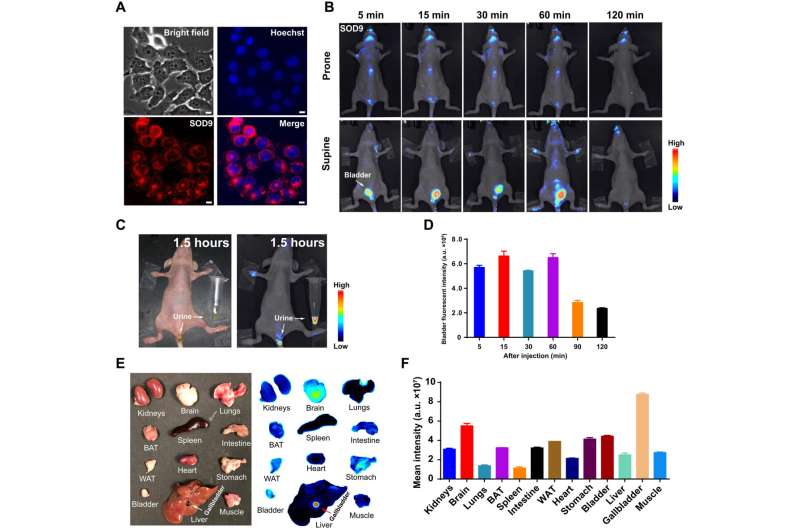
Biocompatibility of the intravenous injection of the dye biomolecules
The researchers studied the biocompatibility of the dye after intravenous injection to a mouse model and investigated its effect on the internal organs via histology. The outcomes highlighted the possibility of using the new fluorescence probe to identify tumors and major organ fluorescence imaging. The work emphasized the best time for fluorescence image-guided tumor surgery to be 60 minutes post-injection and demonstrated how the fast brain accumulation of the dye briefly made it suitable for cranial nerve dynamic imaging. The outcomes highlighted the impact of the dye molecule for brain nerve dynamic monitoring for the first time.
Outlook
In this way, Hao Chen and colleagues designed and developed a series of oxazolone analogs and calculated their optimized geometry. The resulting dye analogs showed a lower bandgap to contribute to a larger Stokes shift approximating 150 to 200 nm greater than traditional fluorescence dyes. The substituents and steric hindrance effects played a key role in influencing the spectroscopic properties of the dyes. The outcomes indicated good optical and pharmacokinetic properties with high signal-to-background ratio, rapid clearance, and low toxicity. The molecules impressively crossed the blood-brain barrier after intravenous injection into mice to provide a strong fluorescence signal to visualize neurons via confocal fluorescence imaging in vivo.
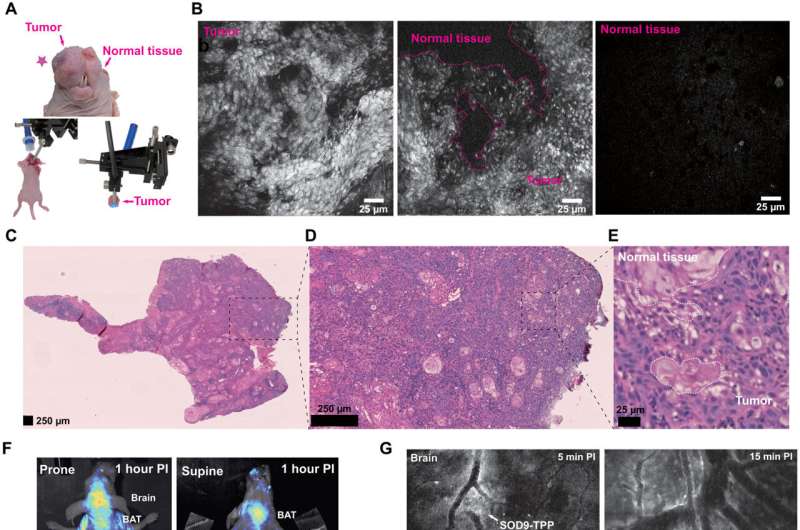
The team further modified the protocol to allow staining of mitochondria in living cells with applications across tumor imaging, fluorescence navigation surgery and confocal endoscopy to identify surgical boundaries and prevent surgical trauma. The new techniques facilitated the analysis of cell morphology in real time, which simplified the process of conventional histological examination with the additional capacity to replace traditional methods of staining such as Hematoxylin and Eosin as well. The dyes are a previously unreported compound that can be used for biomedical applications during fluorescence-guided surgery, with promising properties including high quantum efficiency, low cytotoxicity, rapid excretion and fluorescence imaging.
More information: Hao Chen et al, Bioinspired large Stokes shift small molecular dyes for biomedical fluorescence imaging, Science Advances (2022). DOI: 10.1126/sciadv.abo3289
Guosong Hong et al, Near-infrared fluorophores for biomedical imaging, Nature Biomedical Engineering (2017). DOI: 10.1038/s41551-016-0010
Journal information: Nature Biomedical Engineering , Science Advances
© 2022 Science X Network