Research on bacteria: Electron highway for hydrogen and carbon dioxide storage discovered

In 2013, a team of microbiologists led by Professor Volker Müller from Goethe University Frankfurt discovered an unusual enzyme in a heat-loving (thermophilic) bacterium: the hydrogen-dependent CO2 reductase HDCR. It produces formic acid (formate) from gaseous hydrogen (H2) and carbon dioxide (CO2), and in the process the hydrogen transfers electrons to the carbon dioxide. That makes this HDCR the first known enzyme which can directly utilize hydrogen. In contrast, all enzymes known until then that produce formic acid take a detour: they obtain the electrons from soluble cellular electron transfer agents, which for their part receive the electrons from the hydrogen with the help of other enzymes.
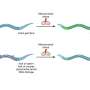
The bacterium Thermoanaerobacter kivui thrives far away from oxygen, for example in the deep sea, and uses CO2 and hydrogen to produce cellular energy. The HDCR of Thermoanaerobacter kivui consists of four protein modules: one that splits hydrogen, one that produces formic acid and two small modules that contain iron sulfur. "It was already clear to us after our discovery that it had to be the two small subunits that transfer the electrons from one module to the other," says Müller. In 2016, the researchers observed that the enzyme forms long filaments. According to Müller they "could see how important this structure was from the fact that filament formation massively stimulates enzyme activity."
The researchers from Goethe University Frankfurt, together with the group led by Dr. Jan Schuller, University of Marburg and LOEWE Centre for Synthetic Microbiology, have now produced a molecular close-up of the enzyme. Through cryo-electron microscopy analysis, Schuller's group has succeeded in determining the HDCR structure at atomic resolution. This made details of the long filaments visible, which the enzyme forms under experimental conditions in the laboratory (in vitro): the filaments' backbone is composed of the two small HDCR subunits, which are arranged together to form a kind of nanowire with thousands of electron-conducting iron atoms. "This is the only enzymatically decorated nanowire discovered so far. On this wire, the hydrogenase module and the formate dehydrogenase module sit like mushroom heads on a cable," explains Schuller.
Helge Dietrich, a doctoral researcher in Volker Müller's group at Goethe University Frankfurt, tested a genetic modification of the small modules that prevented the HDCR filaments from forming. The result: the individual components or monomers were far less active than the filament.
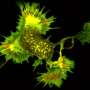
Enzyme monomers arrange themselves into filamentous structures inside bacterial cells too. Professor Ben Engel, a structural cell biologist at the University of Basel, and his team contributed this finding by performing cryo-electron tomography. Using this cutting-edge technique, the researchers discovered something special: "Hundreds of filaments bundle together to form ring-shaped superstructures. These structures are really striking—we informally call them 'portals'," explains Engel. The bundles are evidently anchored in the inner membrane of the bacterial cell and span almost its entire width.
Dr. Ricardo Righetto, senior scientist in Ben Engel's team, analyzed the structure of HDCR filaments within the native bacteria: "Cryo-electron tomography allows us to directly look inside cells at very high resolution. Using this approach, we were really surprised to not only confirm the occurrence of HDCR filaments in the cells but to find they form large bundles attached to membrane".
This structure reveals why the HDCR enzyme is orders of magnitude more efficient than all chemical catalysts and far better than all known enzymes at producing formic acid as a "liquid organic hydrogen carrier" from hydrogen and CO2. Volker Müller: "The hydrogen concentrations in the ecosystem of these bacteria are low, and, in addition, the CO2 and H2 concentrations can switch. Formation of filaments and bundling not only substantially increase the concentration of these enzymes in the cell. The thousands of electron-conducting iron atoms in this 'nanowire' can also store the electrons from hydrogen oxidation intermediately when even just one hydrogen bubble passes by the bacteria."
The team is convinced that not all the enigmas surrounding the HDCR enzyme have yet been solved through the atomic resolution of the structure. Jan Schuller says that "we don't yet know how the wire stores the electrons, why filament formation stimulates enzymatic activity so intensively or how the bundles are anchored in the membrane. We're working on these research questions." But the HDCR's future could be very exciting, believes Volker Müller: "Perhaps one day we'll be able to produce synthetic nanowires which we can use to capture CO2 from the atmosphere. We're also a step closer now to biological hydrogen storage."
The research was published in Nature.
More information: Helge M. Dietrich et al, Membrane-anchored HDCR nanowires drive hydrogen-powered CO2 fixation, Nature (2022). DOI: 10.1038/s41586-022-04971-z
Journal information: Nature
Provided by Goethe University Frankfurt am Main