This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists modify function of common enzymes by hacking genetics, could improve cancer treatments
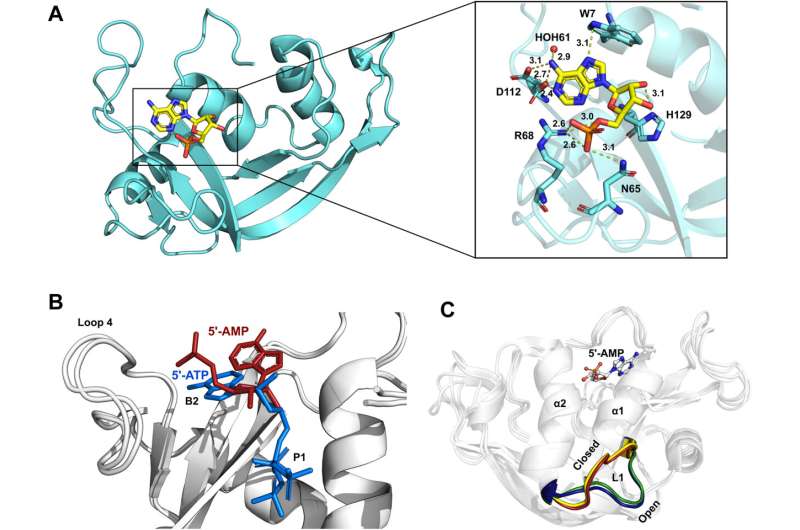
Researchers are using the Canadian Light Source at the University of Saskatchewan to study how enzymes found in all forms of life (called ribonucleases) can be modified to work to our advantage. This technology could have wide-ranging applications, from better cancer treatments and more effective pharmaceuticals to more efficient and environmentally friendly industrial catalysts.
In the human body alone, there are eight active ribonucleases (RNases). These enzymes are secreted by a large variety of different tissues and help manage the messages that come from our DNA. These enzymes are nearly identical in terms of their 3D architecture and molecular makeup, yet they carry out very different functions. For example, some protect us from infections while others contribute to tumor growth.
Prof. Nicolas Doucet and colleagues at the Institut National de la Recherche Scientifique's Armand-Frappier Santé Biotechnologie Research Center, have been studying how to differentiate and modify these enzymes.
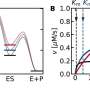
The research team previously discovered that the function of a ribonuclease could be identified by its movements at the molecular level. Now, they have found a way to hack them too.
"By reconstructing the evolutionary ancestor of two enzymes, it allowed us to figure out how mutations have occurred but also how they've influenced specific biological functions throughout evolution," said Doucet. "This provided us with a tool to effectively predict how to transform the activity of an enzyme."
The researchers successfully modified an enzyme so that it became antibacterial and toxic to cells (cytotoxic). This research was recently published in the Journal of Biological Chemistry.
Using the CMCF beamline at the CLS enabled them to analyze enzymes and proteins at an atomic scale.
"This work provides a means to better design inhibitors in the context of drug design or pharmaceutical applications and to design or modify biocatalysts for targeted, specific industrial applications," he explained.
While this area of research is still at an early stage, the new study by Doucet and colleagues shows that engineering our genetics is possible and could bring about exciting advances in medicine and industry.
More information: Thi Thanh Quynh Tran et al, Ancestral sequence reconstruction dissects structural and functional differences among eosinophil ribonucleases, Journal of Biological Chemistry (2024). DOI: 10.1016/j.jbc.2024.107280
Journal information: Journal of Biological Chemistry
Provided by Canadian Light Source