Transforming 'sewer gas' into clean hydrogen fuel
Scientists have found a new chemical process to turn a stinky, toxic gas into a clean-burning fuel.
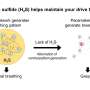
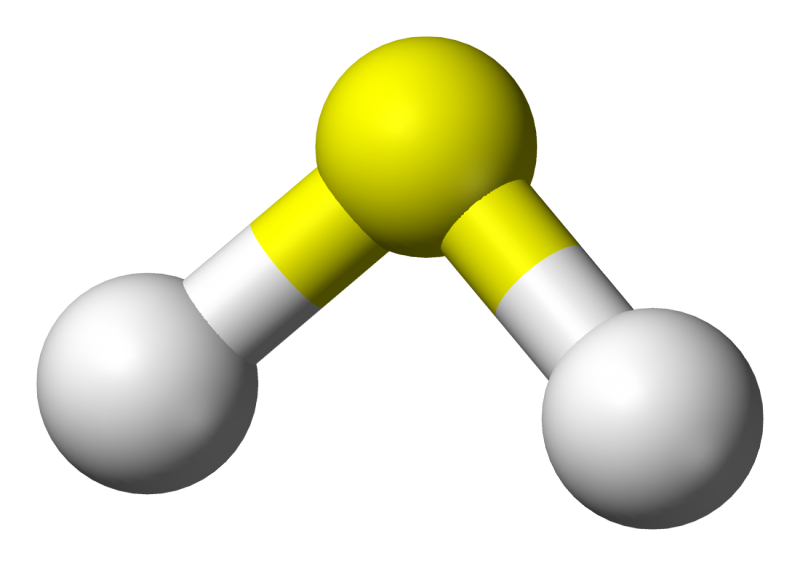
The process, detailed recently in the American Chemical Society journal ACS Sustainable Chemical Engineering, turns hydrogen sulfide—more commonly called "sewer gas"—into hydrogen fuel. Hydrogen sulfide is emitted from manure piles and sewer pipes and is a key byproduct of industrial activities including refining oil and gas, producing paper and mining.
The process detailed in this study uses relatively little energy and a relatively cheap material—the chemical iron sulfide with a trace amount of molybdenum as an additive.
In addition to smelling like rotten eggs, hydrogen sulfide is highly toxic, corroding pipes and harming the health of people who encounter it.
"Hydrogen sulfide is one of the most harmful gases in industry and to the environment," said Lang Qin, a co-author on the study and a research associate in chemical and biomolecular engineering at The Ohio State University. "And because the gas is so harmful, a number of researchers want to turn hydrogen sulfide into something that is not so harmful, preferably valuable."
The study is built on previous work by the same research group using a process called chemical looping, which involves adding metal oxide particles in high-pressure reactors to burn fuels without direct contact between air and fuel. The team first used chemical looping on coal and shale gas to convert fossil fuels into electricity without emitting carbon dioxide into the atmosphere. The initial process used iron oxide to break down the fossil fuels.
The researchers later applied the concept to hydrogen sulfide and invented the SULGEN process, which converts hydrogen sulfide into hydrogen. The researchers found that the pure chemical, iron sulfide, didn't perform well at the large scales needed for industrial use, Qin said. The research team has been trying to identify other inexpensive chemicals that could catalyze that transformation in higher quantities. This study shows that introducing a trace amount of molybdenum into iron sulfide might be an attractive option.
That material is relatively inexpensive and easy to acquire, making it an attractive option for larger-scale operations.
Transforming this toxic gas into hydrogen fuel creates an alternative oil and gas, which are major contributors to climate change, the researchers said.
"It is too soon to tell if our research can replace any of the hydrogen fuel production technologies that are out there," said Kalyani Jangam, lead author of the study and a graduate student in Ohio State's Clean Energy Research Laboratory. "But what we are doing is adjusting this decomposition process and making a valuable product from that."
For this most recent study, the researchers found that molybdenum improves the breakdown of hydrogen sulfide, splitting it into two parts—hydrogen fuel and sulfur.
This work is early in the scientific process—the researchers showed that the process worked in the lab; tests at the industrial level are forthcoming.
"The big picture is we want to solve the harmful gas issue, and we thought that our chemical looping process would allow that," Qin said. "And here, we have found a way to do it in the lab that creates this value-added hydrogen fuel."
The senior author on this paper is Liang-Shih Fan, professor of chemical and biomolecular engineering at Ohio State. Yu-Yen Chen, a former doctoral student in Fan's laboratory, also contributed.
More information: Kalyani Jangam et al, Mo-Doped FeS Mediated H2 Production from H2S via an In Situ Cyclic Sulfur Looping Scheme, ACS Sustainable Chemistry & Engineering (2021). DOI: 10.1021/acssuschemeng.1c03410
Provided by The Ohio State University