Sensor detects whiff of bad breath
Ever wish you could do a quick "breath check" before an important meeting or a big date? Now researchers, reporting in ACS' journal Analytical Chemistry, have developed a sensor that detects tiny amounts of hydrogen sulfide gas, the compound responsible for bad breath, in human exhalations.
According to the American Dental Association, half of all adults have suffered from bad breath, or halitosis, at some point in their lives. Although in most cases bad breath is simply an annoyance, it can sometimes be a symptom of more serious medical and dental problems. However, many people aren't aware that their breath is smelly unless somebody tells them, and doctors don't have a convenient, objective test for diagnosing halitosis. Existing hydrogen sulfide sensors require a power source or precise calibration, or they show low sensitivity or a slow response. Il-Doo Kim and coworkers wanted to develop a sensitive, portable detector for halitosis that doctors could use to quickly and inexpensively diagnose the condition.
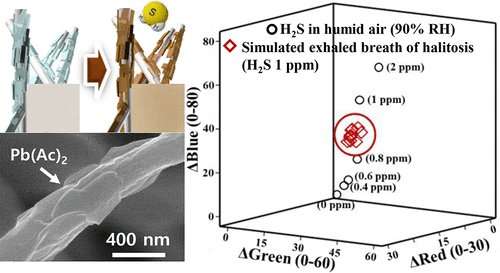
To develop their sensor, the team made use of lead(II) acetate—a chemical that turns brown when exposed to hydrogen sulfide gas. On its own, the chemical is not sensitive enough to detect trace amounts (2 ppm or less) of hydrogen sulfide in human breath. So the researchers anchored lead acetate to a 3-D nanofiber web, providing numerous sites for lead acetate and hydrogen sulfide gas to react. By monitoring a color change from white to brown on the sensor surface, the researchers could detect as little as 400 ppb hydrogen sulfide with the naked eye in only 1 minute. In addition, the color-changing sensor detected traces of hydrogen sulfide added to breath samples from 10 healthy volunteers.
More information: Jun-Hwe Cha et al. Sub-Parts-per-Million Hydrogen Sulfide Colorimetric Sensor: Lead Acetate Anchored Nanofibers toward Halitosis Diagnosis, Analytical Chemistry (2018). DOI: 10.1021/acs.analchem.8b01273
Abstract
Lead(II) acetate [Pb(Ac)2] reacts with hydrogen sulfide to form colored brownish precipitates of lead sulfide. Thus far, in order to detect leakage of H2S gas in industrial sectors, Pb(Ac)2 has been used as an indicator in the form of test papers with a detection limit only as low as 5 ppm. Diagnosis of halitosis by exhaled breath needs sensors able to detect down to 1 ppm of H2S gas. In this work, high surface area and porous Pb(Ac)2 anchored nanofibers (NFs) that overcome limitations of the conventional Pb(Ac)2-based H2S sensor are successfully achieved. First, lead(II) acetate, which melts at 75 °C, and polyacrylonitrile (PAN) polymer are mixed and stirred in dimethylformamide (DMF) solvent at 85 °C, enabling uniform dispersion of fine liquid droplets in the electrospinning solution. During the subsequent electrospinning, Pb(Ac)2 anchored NFs are obtained, providing an ideal nanostructure with high thermal stability against particle aggregation, numerous reactions sites, and enhanced diffusion of H2S into the three-dimensional (3D)-networked NF web. This newly obtained sensing material can detect down to 400 ppb of H2S at a relative humidity of 90%, exhibiting high potential feasibility as a high-performance colorimetric sensor platform for diagnosis of halitosis.
Journal information: Analytical Chemistry
Provided by American Chemical Society