Newly discovered cell type in human skin contributes to inflammatory skin diseases
A team of international scientists and clinical experts have unraveled a new cell type in human skin that contributes to inflammatory skin diseases such as atopic dermatitis (AD) and psoriasis (PSO). Their study findings were published in the Journal of Experimental Medicine in September 2021. The team hails from A*STAR's Singapore Immunology Network (SIgN), in collaboration with the Skin Research Institute of Singapore (SRIS), Singapore's National Skin Centre, Department of Dermatology, Kyoto University Graduate School of Medicine, Japan, and industry partner Galderma.
Chronic inflammatory skin diseases such as AD and PSO are characterized by the presence of an activated T cell subtypes secreting pro-inflammatory cytokines in the skin. This T cell-mediated immune dysregulation is central to the pathogenesis of a wide range of inflammatory skin diseases. Thus, understanding the factors modulating T cell priming and activation in healthy and diseased skin is key to developing effective treatments for these diseases.
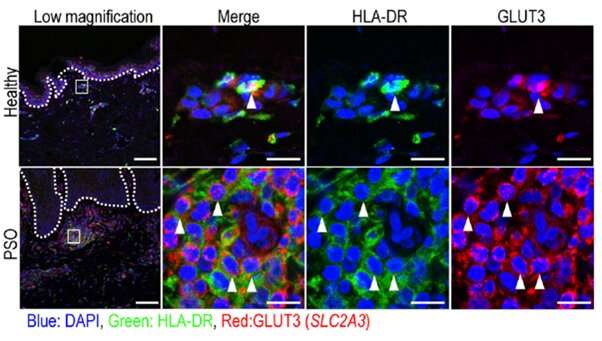
Recently, single-cell RNA sequencing (RNA-seq) approach has been used to analyze immune cells in human skin including dendritic cells (DCs) and macrophages, which are cell populations controlling T cell activation. To address the role of DCs and macrophages in chronic inflammatory skin diseases, the team used a combination of complex approaches (single-cell flow cytometry and RNA-seq of index-sorted cells from healthy and diseased human skin) to generate an unbiased profile/ landscape of DCs and macrophages, and to describe their distinct molecular signatures and proportions in skin lesions of AD and PSO patients.
This uncovered a significant enrichment in the proportion of CD14+ DC3s in PSO lesional skin, where they were one of the major cell types co-expressing IL1B and IL23A, two cytokines essential for PSO pathogenesis. This finding suggests that targeting CD14+ DC3 might represent a novel therapeutic option in the treatment of PSO, and demonstrates the potential for the single-cell myeloid cell landscape database to provide important insights into skin biology in health and disease.
Dr. Florent Ginhoux, senior principal investigator, SIgN, and last author of the study said, "The findings from this study are significant as it will allow the design of new strategies to target or modulate myeloid cell populations for better health outcomes for patients of atopic dermatitis and psoriasis."
"The roles of antigen-presenting cells in the development of inflammatory skin diseases remain unclear. This study clearly revealed the functions of each antigen-presenting cell subset, which is very informative and valuable to understand the pathogenesis of atopic dermatitis and psoriasis. We expect that this study will lead to the design of new treatment for refractory inflammatory skin diseases." said Prof Kenji Kabashima, Adjunct Principal Investigator from SIgN and SRIS.
More information: Satoshi Nakamizo et al, Single-cell analysis of human skin identifies CD14+ type 3 dendritic cells co-producing IL1B and IL23A in psoriasis, Journal of Experimental Medicine (2021). DOI: 10.1084/jem.20202345
Journal information: Journal of Experimental Medicine