How a COVID-19 infection changes blood cells in the long run

Using real-time deformability cytometry, researchers at the Max-Planck-Zentrum für Physik und Medizin in Erlangen were able to show for the first time: COVID-19 significantly changes the size and stiffness of red and white blood cells—sometimes over months. These results may help to explain why some affected people continue to complain of symptoms long after an infection (long COVID).
Shortness of breath, fatigue and headaches: Some patients still struggle with the long-term effects of a severe infection by the SARS-CoV-2 coronavirus after six months or more. This post-COVID-19 syndrome, also called long COVID, is still not properly understood. What is clear is that—during the course of the disease—often blood circulation is impaired, dangerous vascular occlusions can occur and oxygen transport in is limited. These are all phenomena in which the blood cells and their physical properties play a key role.
To investigate this aspect, a team of scientists led by Markéta Kubánková, Jochen Guck, and Martin Kräter from the Max-Planck-Zentrum für Physik und Medizin, the Max Planck Institute for the Science of Light (MPL), the Friedrich Alexander University Erlangen-Nuremberg and the German Center for Immunotherapy measured the mechanical states of red and white blood cells. "We were able to detect clear and long-lasting changes in the cells—both during an acute infection and even afterwards," reports Professor Guck, currently managing director of MPL. The research group has now published their results in the renowned journal Biophysical Journal.
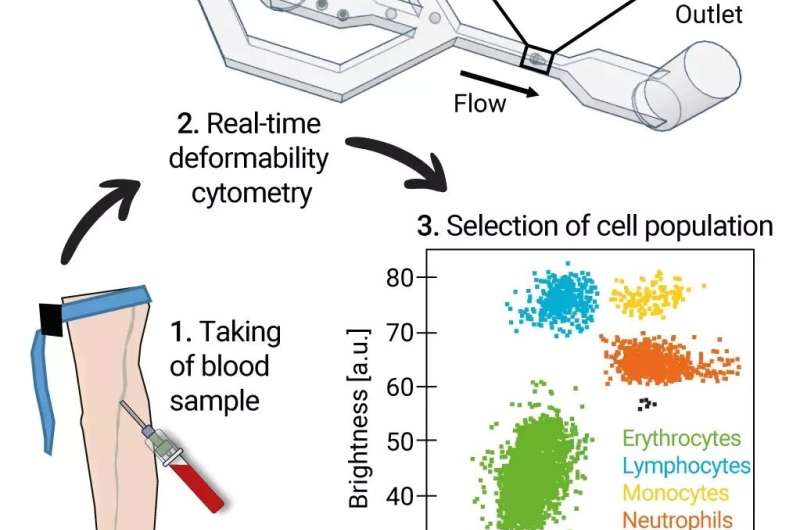
To analyze the blood cells, they used a self-developed method called real-time deformability cytometry (RT-DC), which has recently been recognized with the prestigious Medical Valley Award. In this method, the researchers send the blood cells through a narrow channel at high speed. In the process, the leukocytes and erythrocytes are stretched. A high-speed camera records each of them through a microscope, and custom software determines which cell types are present, and how big and deformed they are. Up to 1000 blood cells can be analyzed per second. The advantage of the method: It is fast and the cells do not have to be labeled in a laborious procedure.
The method could help as an early warning system to detect future pandemics by unknown viruses
The biophysicists from Erlangen examined more than four million blood cells from 17 patients acutely ill with COVID-19, from 14 people who had recovered and 24 healthy people as a comparison group. They found that, for example, the size and deformability of the red blood cells of patients with the disease deviated strongly from those of healthy people. This indicates damage to these cells and could explain the increased risk of vascular occlusion and embolisms in the lungs. In addition, the oxygen supply, which is one of the main tasks of the erythrocytes, may be impaired in infected persons. Lymphocytes (one type of white blood cell responsible for the acquired immune defense) were in turn significantly softer in COVID-19 patients, which typically indicates a strong immune reaction. The researchers made similar observations for neutrophil granulocytes, another group of white blood cells involved in the innate immune response. These cells even remained drastically altered seven months after the acute infection.
"We suspect that the cytoskeleton of immune cells, which is largely responsible for cell function, has changed," explains Markéta Kubánková, first author of the research article. In her view, real-time deformability cytometry has the potential to be used routinely in the diagnosis of COVID-19—and even to serve as an early warning system against future pandemics caused by as yet unknown viruses.
More information: Markéta Kubánková et al, Physical phenotype of blood cells is altered in COVID-19, Biophysical Journal (2021). DOI: 10.1016/j.bpj.2021.05.025
Journal information: Biophysical Journal
Provided by Max Planck Society