Researchers discover SARS-CoV-2 inhibitors
A research team of pharmacists at the University of Bonn has discovered two families of active substances that can block the replication of the SARS-CoV-2 coronavirus. The drug candidates are able to switch off the the key enzyme of the virus, the so-called main protease. The study is based on laboratory experiments. Extensive clinical trials are still required for their further development as therapeutic drugs. The results have now been published in the journal Angewandte Chemie.
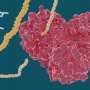
In order for the SARS-CoV-2 coronavirus to replicate, it relies on the main protease as a key enzyme. The virus first has its genome translated from RNA into a large protein strand. The viral main protease then cuts this protein chain into smaller pieces, from which the new virus particles are formed. "The main protease is an extremely promising starting point for coronavirus drug research," says Prof. Dr. Christa E. Müller of the Pharmaceutical Institute at the University of Bonn. "If this enzyme is blocked, viral replication in the body's cells is stopped." The researcher is a member of the Transdisciplinary Research Area "Life and Health" at the University of Bonn.
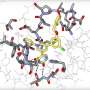
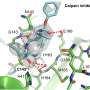
The pharmaceutical chemists designed a large number of potential inhibitors based on the structure of the main protease and the mechanism by which the important virus-replicating enzyme works. "A suitable inhibitor must bind sufficiently tightly to the main protease to be able to block its active site," says Prof. Dr. Michael Gütschow, who heads an independent research group on such inhibitors at the Pharmaceutical Institute of the University of Bonn.
Fluorescent test system
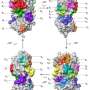
Then the experimental phase began. The researchers developed a new test system for high-throughput screening. They offered the main protease a substrate to which a reporter molecule was coupled. When the protease catalytically cleaved this coupling, the fluorescence of the product was measurable. However, if a simultaneously administered inhibitor successfully blocked the activity of the protease, there was no fluorescence. "For most of the test compounds, we observed no enzyme inhibition. But on rare occasions in our comprehensive tests, fluorescence was suppressed: These were the hits we had hoped for in our search for inhibitors of the viral protease," reports Gütschow.
Like chewing gum at the catalytic center
The researchers' high-throughput screening showed two classes of drugs that appeared to be particularly promising. Customized compounds of both classes were then newly synthesized. They stick to the main protease like chewing gum and block the crucial catalytic center, which prevents the main protease from preparing the virus replication. "Some of the compounds even have another effect," Müller reports. "They also inhibit a human enzyme that helps the virus enter body cells."
The participants contributed very different expertise to the study. "Only through great collaboration have we been able to design, synthesize and biochemically characterize suitable drug candidates," says Gütschow. "The best compounds represent promising lead structures for drug development," says Müller. However, extensive clinical trials have yet to prove whether these candidates also inhibit SARS coronavirus-2 replication in humans, Gütschow adds.
More information: Julian Breidenbach et al. Targeting the Main Protease of SARS‐CoV‐2: From the Establishment of High Throughput Screening to the Design of Tailored Inhibitors, Angewandte Chemie International Edition (2021). DOI: 10.1002/anie.202016961
Journal information: Angewandte Chemie International Edition , Angewandte Chemie
Provided by University of Bonn