Antimicrobial peptides accelerate lipid transport in the bacterial membrane
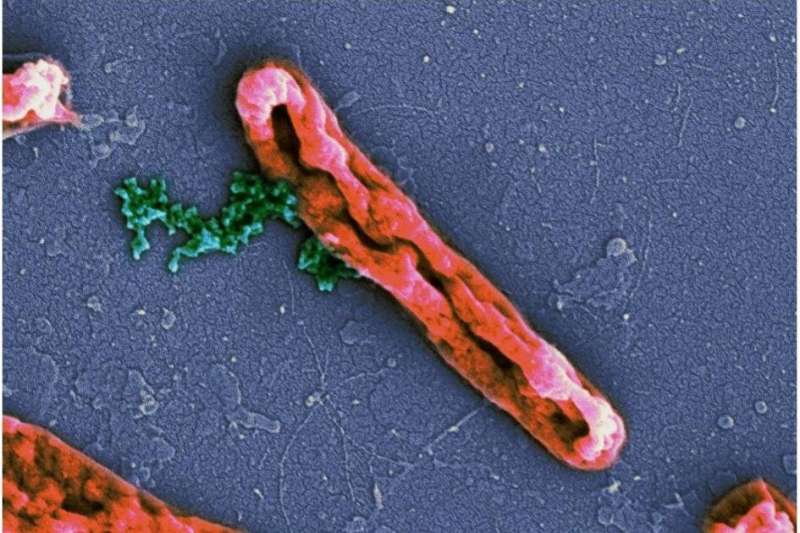
New insight into how antimicrobial peptides destroy the membrane may help in designing better drugs against multi-drug resistant bacterial infections. Now that we are in the midst of a pandemic, it is easy to forget the perhaps even greater threat from the antibiotic-resistant bacteria.
After Alexander Fleming's first discovery of penicillin, new types of antibiotics were constantly being developed to defend us against dangerous bacteria. Now we often hear about bacteria that cannot be killed and are almost impossible to get rid of.
"Currently there is limited research focus into finding and developing new antibiotics, especially in the medicinal industry. The development has sort of stagnated, and we often see that resistance mechanisms already exist or develop shortly after identification of a new drug," says Josefine Eilsø Nielsen.
She has just received her doctorate from the Department of Chemistry at the University of Oslo and is one of those looking for other alternatives than antibiotics in the ongoing war against bacteria.
"In my Ph.D. project I have studied a class of compounds referred to as antimicrobial peptides. These peptides are found amongst others humans and animals," Nielsen says to Titan.uio.no.
Peptides consist of amino acids, the same building blocks as proteins, but they are much smaller than the proteins.
What happens on a molecular level?
Antimicrobial peptides can, as the name implies, destroy microbes. Microbes are not only bacteria, but also include other microscopic organisms such as viruses and fungi.
"We are by far the first people to have these ideas. There is significant research effort into the field of antimicrobial peptides, but there are few drugs that have reached the clinical marked," Nielsen says.
She has previously completed a master of Pharmacy at the Department of Pharmacy and worked at the Norwegian Medicines Agency, before starting her chemistry Ph.D. studies.
The drug Colistin is based on an antimicrobial peptide, but due to possible side effects it is not used until other treatments have failed.
"This is an antibiotic that is used as a last resort treatment in patients with multi-drug resistant bacterial infections."
In order to move forward, it is necessary to find out how these peptides actually work, all the way down to the molecular level where Nielsen is operating.
"If we can understand what happens on a molecular level, this will hopefully help guide collaborators in the design of better drug candidates."
Messing with the balance in the membrane
Nielsen has investigated what the antimicrobial peptides do to the membrane, the protective layer that surrounds the bacterial cell. It has been established that the peptides can destroy the membrane, but not exactly how this happens.
"Previous research has suggested that these peptides act by forming defined channels in the bacterial membrane, and thereby altering the transport of fluids and ions though the membrane, causing bacterial cell death," Nielsen says.
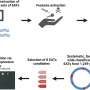
She found no evidence of such channels when she examined an artificial bacterial membrane in more detail using X-rays and neutron scattering.
"The bacterial membrane is composed of a bilayer, and there is spontaneous transport of lipids between the outer and inner layer, but this process is slow. This changes upon addition of antimicrobial peptide. Then the transport of lipids between inner and outer layer is accelerated significantly," Nielsen says.
"We hypothesize that these changes in lipid transport in the cell membrane might potentially be enough to kill the bacterial cell."
In nature the lipid transport and lipid composition in each layer is extremely well regulated and balanced.
"When these antimicrobial peptides messes with the balance, this might be enough to kill the bacterium," Nielsen says.
Viruses, fungi, Alzheimer's and diabetes
If it is sufficient to create this unbalance in lipid transport, it will be easier to design smaller and less toxic drugs candidates, without the necessity of more complex molecules that are able to form defined membrane channels, as prior theories would suggest.
"Because these antimicrobial peptides act on the membrane itself and therefore is less dependent on transporting into the bacterial cell, it is less likely that resistance mechanisms against these drugs will develop," Nielsen says.
There is still a long way to go before we have new drugs that can attack bacteria, but knowledge of what is going on has come a step further. Nielsen will continue to research these peptides when she now enters a postdoctoral position at the Stanford University.
"What is really exciting is that these peptides not only affect different types of bacteria, but also fungi, parasites and viruses. They have a very broad effect," Nielsen says.
More information: Josefine Eilsø Nielsen et al. Beyond structural models for the mode of action: How natural antimicrobial peptides affect lipid transport, Journal of Colloid and Interface Science (2020). DOI: 10.1016/j.jcis.2020.08.094
Provided by University of Oslo