Research improves ion evaporation theories and performance of electrospray applications
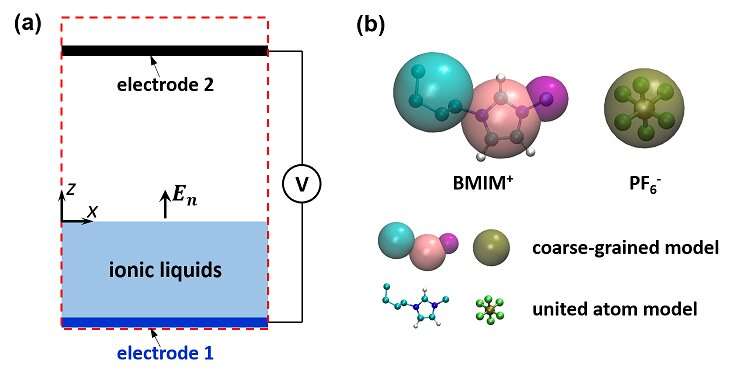
Electrospray of room temperature ionic liquids (RTILs), which are solvent-free electrolytes with easily tailored ions, is emerging as a powerful tool in diverse fields. In particular, electrosprays of RTILs operating in the pure-ion mode have attracted significant attention recently. However, despite intensive technological development, these electrosprays have yet to achieve the robustness and effectiveness demanded by their applications. Mechanisms behind these electrosprays remain poorly understood.
Dr. Jiang Xikai from the Institute of Mechanics of the Chinese Academy of Sciences (CAS) and his collaborators studied the electrospray of RTILs. By using MD simulations, they investigated electric-field-driven ion emission from the free surface of a planar RTIL film. They calculated ion emission rate as a function of the electric field normal to the RTIL/vacuum surface and found that their relationship agrees with predictions from classical ion evaporation theories.
"This is the very first time that the classical scaling law in ion evaporation theories is recovered in simulations," said Dr. Jiang, corresponding author of this study.
The composition of emitted ions includes monomers and dimers. It was found that the monomer has to move across two barriers before emission: one above the RTIL/vacuum surface, which agrees with classical ion evaporation theories; one underneath the surface due to the unique structure of RTIL/vacuum surface as revealed by simulations. The fraction of dimers was found to depend on the external field and ion-ion interactions.
To understand how different species form, the researchers further performed replica exchange molecular dynamics simulations and identified four metastable states of the emitting ion near the liquid film. These metastable states strongly affect the composition of ion emission.
Fundamental insights revealed in this study form the basis to improve ion evaporation theories and guide rational selection of RTILs to achieve desired ion emission characteristics.
More information: Fei Zhang et al. Electric-Field-Driven Ion Emission from the Free Surface of Room Temperature Ionic Liquids, The Journal of Physical Chemistry Letters (2021). DOI: 10.1021/acs.jpclett.0c03335
Journal information: Journal of Physical Chemistry Letters
Provided by Chinese Academy of Sciences