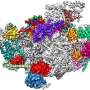
Human ribosome assembly has no counterparts in simpler model organisms
Ribosomes synthesize all the proteins in cells. Studies mainly done on yeast have revealed much about how ribosomes are put together, but an Ludwig-Maximilians-Universitaet (LMU) in Munich team now reports that ribosome assembly in human cells requires factors that have no counterparts in simpler model organisms.
In every cell, hundreds of thousands of intricate molecular machines called ribosomes fabricate new proteins, extending each growing chain at a rate of a few amino acids per second. Not surprisingly, therefore, the construction of these vital protein factories is itself a highly complex operation, in which more than 200 assembly factors are transiently involved. Mature ribosomes are made up of approximately 80 proteins and four ribosomal RNAs. But how these constituents are assembled in the correct order to yield a functional ribosome is still not fully understood. Moreover, most of our knowledge of the process comes from studies carried out on model organisms like bacteria and yeast, and may not necessarily be applicable to the cells of higher organisms. Researchers led by Professor Roland Beckmann (Gene Center, LMU Munich) have now uncovered new details of the crucial steps in the maturation of ribosomes in human cells.
Active ribosomes consist of two separately assembled particles, which differ in size and interact with each other only after the first steps in protein synthesis have taken place on the smaller of the two (in human cells, the '40S subunit'). Beckmann's team has used cryo-electron microscopy to determine the structures of several precursors of the 40S subunit isolated from human cells and follow the course of its maturation. "This study follows on from an earlier project, in which we obtained initial insights into the process," says Michael Ameismeier. He is a doctoral student in Beckmann's team and lead author of the new report, which is concerned with the final steps in the assembly of the small subunit.
At this late stage in the process, one end of the ribosomal RNA associated with the small particle protrudes from the body of the immature subunit. The last step in the maturation of the 18S subunit consists in the removal of this now superfluous segment. To ensure that this reaction does not occur prematurely, the enzyme responsible—NOB1—is maintained in an inactive state until it is required. The new study shows that the activation of NOB1 is preceded by a conformational change that results in the detachment of a binding partner from the enzyme. This in turn triggers a structural rearrangement in NOB1 itself, which enables the enzyme to snip off the protruding rRNA segment. "The activation of NOB1 is coordinated by another enzyme," Ameismeier explains. Together with a protein we have discovered—which is not found in yeast—the latter enzyme inserts like a wedge into the maturing 40S subunit, and this facilitates the decisive conformational change in NOB1."
The authors have also shown that yet another protein not found in yeast plays an (as yet) enigmatic role in the maturation of the 40S subunit. "This demonstrates the importance of considering the human system separately from other experimental models," says Beckmann. Use of the evolutionarily simpler yeast system is sufficient for a basic understanding of the process. But certain pathological syndromes have been linked to errors in ribosomal biogenesis in humans, which provides an obvious rationale for the study of ribosomal assembly in human cell systems.
More information: Michael Ameismeier et al, Structural basis for the final steps of human 40S ribosome maturation, Nature (2020). DOI: 10.1038/s41586-020-2929-x
Journal information: Nature
Provided by Ludwig Maximilian University of Munich