Scientists propose redox mechanism for water-gas shift reaction
The water-gas shift (WGS) reaction (CO+H2O→CO2+H2) is critical for producing high-purity hydrogen for ammonia and methanol synthesis, as well as in fuel cell applications.
Via the concept of single-atom catalysts (SACs), scientists can now understand the chemical bond and electronic structure between metal and support at an atomic scale.
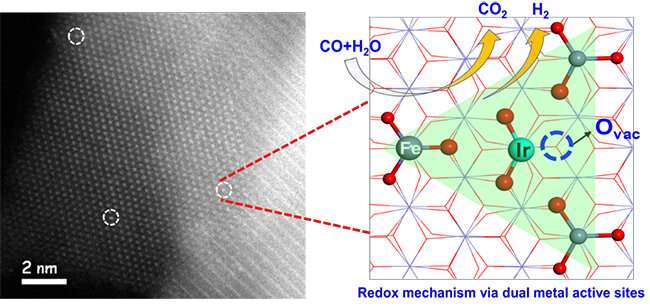
A research group led by Prof. Wang Xiaodong and Prof. Zhang Tao from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), in collaboration with Prof. Li Jun from Tsinghua University, have proposed a redox mechanism with synergetic dual metal active sites for the WGS reaction on a Ir1/FeOx SAC.
The results were published in Angewandte Chemie International Edition.
The scientists found that water easily dissociated to OH* on the Ir1 single atom and H* on the nearby O atom bonded with a Fe site. The adsorbed CO on Ir1 reacted with the adjacent O atom to produce CO2. Then the formation of H2 became feasible due to migration of H from adsorbed OH* toward Ir1 and its subsequent reaction with another H*.
They proposed a new pathway of a redox mechanism via synergetic dual metal active sites between Ir1 and Fe species (DMAS) during the WGS reaction, which demonstrated the sequential production of CO2 and H2.
The elucidation of the catalytic mechanism involving this kind of dual metal active site might provide insights for understanding the catalytic mechanism of metal catalysts with reducible supports, thus contributing to the rational design of new and active dual-site single-atom and single-cluster catalysts.
More information: Jin‐Xia Liang et al. Dual Metal Active Sites in an Ir 1 /FeO x Single‐Atom Catalyst: A Redox Mechanism for the Water‐Gas Shift Reaction, Angewandte Chemie International Edition (2020). DOI: 10.1002/anie.201914867
Journal information: Angewandte Chemie International Edition
Provided by Chinese Academy of Sciences