Both Sn and Zn single-atoms on CuO catalyst synergistically promote dimethyldichlorosilane synthesis

Because of their maximum atom-utilization efficiency and unique catalytic properties, single-atom catalysts (SACs) have sparked intense interests in recent years. However, most of the reported SACs are limited to single-site active components, with rare reports on catalyst promoters in their single forms. Because promoters are essential components in many industrial catalysts, the exploration of the preparation of single-site promoters should be of great interest in catalysis, both in fundamentals and application research. Similar to SACs, these single sited promoters have structural simplicity and homogeneity, and its synergistic effect on the catalytic reaction should be unique but yet clarified.
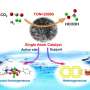
In a recent article published in the Beijing-based National Science Review, scientists at the General Research Institute for Nonferrous Metal (GRINM) in Beijing, China, GRIPM Advanced Materials Co., Ltd. In Beijing, China and Institute of Process Engineering, Chinese Academy of Sciences in Beijing, China, have designed and synthesized atomically dispersed co-promoters of Sn and Zn on the CuO surface. As demonstrated, this catalyst exhibited greatly enhanced promoting effect in the industrially important Rochow reaction for dimethyldichlorosilane synthesis. Also, for the first time, the synergistic promotion mechanism has also been revealed.
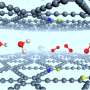
The authors employed a facile hydrothermal method to synthesize Sn1/CuO with a large number of surface Cu vacancies. Furthermore, they investigated the structure of this new catalyst employing various characterization methods and proved the successful uploading of the two single-site promoters. The XPS data gave direct evidence that there is a strong interaction between Sn and Zn atoms. "After incorporation with Zn atoms, the binding energy of Cu 2p3/2 peak shifts to a lower-energy side in comparison with that of CuO, and this shift is observed obviously in 0.1Zn1-Sn1/CuO, indicating an increase of the electron density on the Cu atoms with the coexistence of Sn and Zn atoms," they state. Direct experimental results showed that these defect sites generated by incorporating single-site Sn could further stabilize single-site Zn (see below figure). "Density functional theory (DFT) calculations also show that on Sn-doped CuO(110) surface, the formation energy of Cu vacancy is 0.78 eV lower than that on the clean CuO(110), which indicates it is easier to form Cu vacancies in the Sn-doped surface," they add. The calculation results also support that Zn prefers to fill in the nearby Cu vacancies caused by Sn doping to form Sn-Zn pairs.
Comparing with the conventional catalysts with promoters in the form of nanoparticles, this novel Zn1-Sn1/CuO catalyst has much higher activity, selectivity, and stability in the synthesis of dimethyldichlorosilane via the industrially important Rochow reaction. The enhanced catalytic performance is attributed to the synergistic interaction between single-site Sn and Zn co-promoters, which leads to the change in the electronic structure of CuO and thus promotes the adsorption of reactant molecules.
"These single-sited promoters not only help to elucidate their real promotion mechanism in catalytic reaction, but also open up a new path to optimize catalyst performance," they state in an article titled "Single-atom Sn-Zn pairs in CuO catalyst promote dimethyldichlorosilane synthesis."
This work got the supports of Dr. Wenxin Chen in Beijing Institute of Technology, China; Prof. Jianmin Ma in Hunan University, China; Prof. Ziyi Zhong in Guangdong Technion Israel institute of Technology (GTIIT), China; and Prof. Yadong Li in Tsinghua University, China.
"This work provides a new understanding of the synergistic effect among various promoters and will offer avenues to the design of new co-promoters in catalysts for industrial reactions," they believe.
More information: Qi Shi et al, Single-atom Sn-Zn Pairs in CuO Catalyst Promote Dimethyldichlorosilane Synthesis, National Science Review (2019). DOI: 10.1093/nsr/nwz196
Provided by Science China Press