Liquid metals break down organic fuels into ultra-thin graphitic sheets
For the first time, researchers at the University of New South Wales (UNSW), Sydney, Australia, have demonstrated the synthesis of ultra-thin graphitic materials at room temperature using organic fuels. These fuels can be as simple as basic alcohols such as ethanol. Graphitic materials, such as graphene, are ultra-thin sheets of carbon compounds are promising for battery storage, solar cells, touch panels and fillers for polymers. The results are published in Advanced Materials.
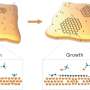
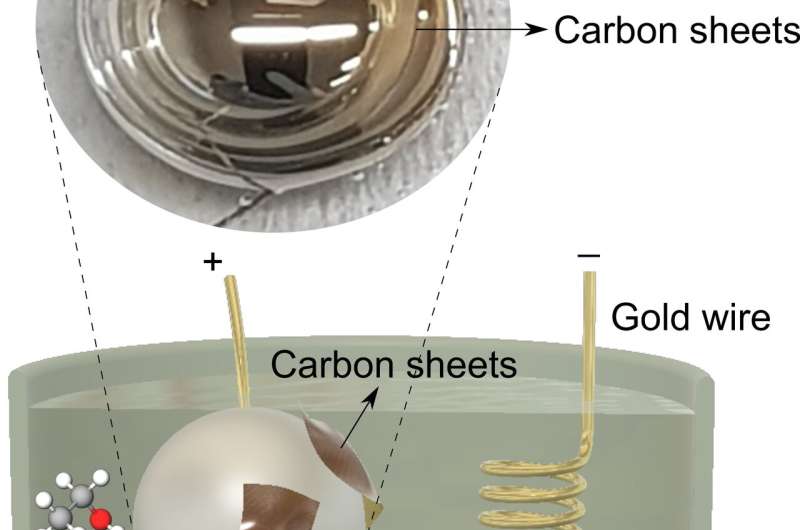
The researchers electrochemically synthesized ultra-thin carbon-based materials on the surface of liquid metals at room temperature. Previously, other researchers had accomplished electro-formation of such carbon-based materials only by transferring sheets onto the electrodes or electrode exfoliation of naturally occurring carbon crystals from mines.
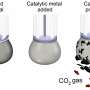
"Using gallium liquid metal, we could catalytically break down the fuels and form carbon-carbon bonds (the base of graphitic sheets) from organic fuels at room temperature. The ultra-smooth surface of liquid metals could then template atomically thin carbon-based sheets. Removal of these sheets was easy, as they do not stick to the liquid metal surface," says Prof Kalantar-Zadeh, the project lead and the director of the Centre for Advanced Solid and Liquid based Electronics and Optics (CASLEO) at UNSW.
"It is simple. Why has room temperature electro-synthesis of two-dimensional graphitic materials not been achieved before? We cannot offer a definitive answer. Perhaps disregarding ultra-catalysts such as liquid metals and too much emphasis on solid electrodes which are inherently not smooth," said Dr. Mohannad Mayyas, the first author of the paper.
More information: Mohannad Mayyas et al. Liquid‐Metal‐Templated Synthesis of 2D Graphitic Materials at Room Temperature, Advanced Materials (2020). DOI: 10.1002/adma.202001997
Journal information: Advanced Materials
Provided by University of New South Wales