A dual-purpose metabolic switch

Compounds called inositol diphosphates are cellular signaling "codes" involved in multiple processes ranging from phosphate sensing to DNA metabolism.
John York, Ph.D., and colleagues have demonstrated that the evolutionarily conserved gene product Vip1 is capable of both synthesizing and destroying inositol diphosphates.
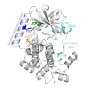
The researchers showed that Vip1 has two independent catalytic domains tethered together: a kinase domain that adds phosphate groups and a pyrophosphatase domain that removes them.
They provided atomic resolution structures of inositol diphosphate products to define the selective site of Vip1 action. Perturbation of kinase or pyrophosphatase activities had differential effects on vacuolar morphology and osmotic responses.
The findings, reported in the Proceedings of the National Academy of Sciences, demonstrate that Vip1 represents a rare class of bifunctional enzyme. This single gene product is a key metabolic switch critical for nutrient and regulatory responses, the authors conclude.
More information: D. Eric Dollins et al. Vip1 is a kinase and pyrophosphatase switch that regulates inositol diphosphate signaling, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1908875117
Journal information: Proceedings of the National Academy of Sciences
Provided by Vanderbilt University