Large dipole moment induced wide-spectrum bismuth chromate for efficient photocatalytic performance
The conversion and utilization of solar energy for chemical fuel production and environmental remediation through artificial photocatalysis have been recognized to be an ideal route to address the critical energy and environmental concerns. The full utilization of solar light is a great challenge for achieving sufficient efficiency in practical applications, and narrowing the bandgap of a photocatalyst weakens the driving force for redox reactions, especially water oxidation and pollutant degradation, because these reactions involve a complicated multi-electron process. Therefore, the development of a wide-spectrum, responsive and highly efficient photocatalysts for water oxidation and pollutant degradation is a critical issue to be addressed at present.
Bi-based oxometallate materials, such as BiVO4¬, Bi2WO6, Bi2MoO6, etc., have been widely studied as visible-light active photocatalysts and exhibit excellent photocatalytic performance in water oxidation and pollutant degradation, which is mainly benefiting from their sufficiently deep valence band position. In this work, a wide-spectrum responsive Bi8(CrO4)O11 nanorod photocatalyst was successfully constructed. Owing to the hybridization of Cr 3d with O 2p orbitals shifts conduction band minimum down, Bi8(CrO4)O11 allows its absorption up to the entire visible region (~678 nm) with a theoretical solar spectrum efficiency of 42.0%. And its VB of 1.95 eV (vs. NHE pH=7) is more positive than the oxidation potential of OH-/O2, which indicates that the photogenerated holes of Bi8(CrO4)O11 nanorod photocatalyst possess extremely strong oxidation capability.
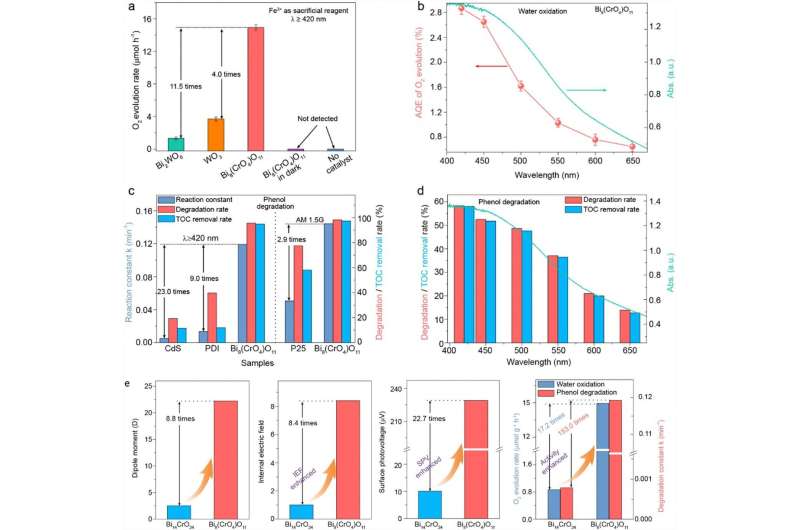
As shown in Figure 1a, Bi8(CrO4)O11 exhibited exceedingly superior photocatalytic water oxidation performance, and its average O2 evolution rate reached 14.94 μmol h-1, about 11.5 and 4.0 times higher than that of Bi2WO6 nanosheets and commercial WO3 nanoparticles. Besides, it consequently achieved a considerable apparent quantum efficiency (AQE) 2.87% at 420 nm, even 0.65% at 650 nm (Figure 1b), higher than many reported wide-spectrum driven photocatalysts. Most noticeably, its excellent activity is also manifested in photocatalytic degradation of phenol. Its degradation reaction constant could reach 0.119 min-1, about 22.5 and 8.8 times higher than CdS nanowires and PDI supramolecular photocatalysts, respectively (Figure 1c). Even its degradation activity is not inferior to P25 TiO2 under simulated sunlight, about 2.9 times higher than the latter.
Remarkably, Bi8(CrO4)O11 also presented extremely strong mineralization ability, which almost enables the simultaneous degradation and complete mineralization for phenol. The total organic carbon removal rates of phenol over Bi8(CrO4)O11 under visible light and simulated sunlight is 94.8% (degradation rate: 95.5%) and 97.3% (degradation rate: 98.1%) in 0.5 h, respectively, while that of CdS, PDI and P25 are significantly lower than their corresponding degradation rates. Even under 650 nm red light irradiation, Bi8(CrO4)O11 is still able to simultaneously degrade and completely mineralize phenol (Figure 1d), and few wide-spectrum driven photocatalysts can achieve that.
Besides, the dipole moments of Bi8(CrO4)O11 was calculated to 22.32 Debye (D), which results in a giant internal electric field (IEF). As shown in Figure 1e, compared to Bi14CrO24, Bi8(CrO4)O11 with a greater dipole showed a significantly higher IEF, charge separation efficiency and photocatalytic performance. Therefore, as illustrated in Scheme 1, the large crystal dipole of Bi8(CrO4)O11 induces a giant IEF, which accelerates the rapid separation of photogenerated electron-hole pairs and exponentially enhances its photocatalytic performance. Most importantly, based on the above mechanism, many more efficient photocatalysts can be designed successfully by regulating the crystal dipole.
More information: Xianjie Chen et al, Large dipole moment induced efficient bismuth chromate photocatalysts for wide-spectrum driven water oxidation and complete mineralization of pollutants, National Science Review (2019). DOI: 10.1093/nsr/nwz198
Provided by Science China Press